04/01/2023 | EAEU Decisions
Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 92 (as amended on February 28, 2023)
Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94
«On technological documents regulating information interaction in the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices»
(together with the «Rules for information interaction in the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices», «Regulation on information interaction between the competent authorities of the Member States of the Eurasian Economic Union and the Eurasian Economic Commission in the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices», «Description of formats and structures of electronic documents and data used for the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices», «Procedure for joining the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices»)
BOARD OF THE EURASIAN ECONOMIC COMMISSION
DECISION
of August 30, 2016 No. 94
ON TECHNOLOGICAL DOCUMENTS REGULATING INFORMATION INTERACTION IN THE IMPLEMENTATION, BY MEANS OF THE INTEGRATED INFORMATION SYSTEM OF FOREIGN AND MUTUAL TRADE, OF THE COMMON PROCESS «FORMATION, MAINTENANCE AND USE OF THE UNIFIED INFORMATION DATABASE FOR MONITORING THE SAFETY, QUALITY AND EFFICACY OF MEDICAL DEVICES»
In accordance with paragraph 30 of the Protocol on Information and Communication Technologies and Information Interaction within the Eurasian Economic Union (Annex No. 3 to the Treaty on the Eurasian Economic Union of May 29, 2014) and guided by Decision of the Board of the Eurasian Economic Commission of November 6, 2014 No. 200, the Board of the Eurasian Economic Commission has decided:
1. To approve the attached:
Rules for information interaction in the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices»;
Regulation on information interaction between the competent authorities of the Member States of the Eurasian Economic Union and the Eurasian Economic Commission in the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices»;
Description of formats and structures of electronic documents and data used for the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices»;
Procedure for joining the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices».
2. To establish that the development of technical schemas for the structures of electronic documents and data provided for by the Description approved by this Decision, and the ensurance of their placement in the register of structures of electronic documents and data used in the implementation of information interaction in the integrated information system of foreign and mutual trade, shall be carried out by the department of the Eurasian Economic Commission whose competence includes the coordination of work on the creation and development of the integrated information system of the Eurasian Economic Union.
3. This Decision shall enter into force after 30 calendar days from the date of its official publication.
Chairman of the Board
of the Eurasian Economic Commission
T. SARGSYAN
Approved
by Decision of the Board
of the Eurasian Economic Commission
of August 30, 2016 No. 94
RULES
FOR INFORMATION INTERACTION IN THE IMPLEMENTATION, BY MEANS OF THE INTEGRATED INFORMATION SYSTEM OF FOREIGN AND MUTUAL TRADE, OF THE COMMON PROCESS «FORMATION, MAINTENANCE AND USE OF THE UNIFIED INFORMATION DATABASE FOR MONITORING THE SAFETY, QUALITY AND EFFICACY OF MEDICAL DEVICES»
I. General Provisions
1. These Rules are developed in accordance with the following acts comprising the law of the Eurasian Economic Union (hereinafter - the Union):
Treaty on the Eurasian Economic Union of May 29, 2014;
Agreement on Common Principles and Rules for Circulation of Medical Devices (Medical Equipment and Medical Materials) within the Eurasian Economic Union of December 23, 2014;
Decision of the Supreme Eurasian Economic Council of December 23, 2014 No. 109 «On the Implementation of the Agreement on Common Principles and Rules for Circulation of Medical Devices (Medical Equipment and Medical Materials) within the Eurasian Economic Union»;
Decision of the Council of the Eurasian Economic Commission of February 12, 2016 No. 30 «On Approval of the Procedure for the Formation and Maintenance of the Information System in the Field of Circulation of Medical Devices»;
Decision of the Board of the Eurasian Economic Commission of November 6, 2014 No. 200 «On Technological Documents Regulating Information Interaction in the Implementation of Common Processes by Means of the Integrated Information System of Foreign and Mutual Trade»;
Decision of the Board of the Eurasian Economic Commission of January 27, 2015 No. 5 «On Approval of the Rules for Electronic Data Exchange in the Integrated Information System of Foreign and Mutual Trade»;
Decision of the Board of the Eurasian Economic Commission of April 14, 2015 No. 29 «On the List of Common Processes within the Eurasian Economic Union and the Introduction of an Amendment to Decision of the Board of the Eurasian Economic Commission of August 19, 2014 No. 132»;
Decision of the Board of the Eurasian Economic Commission of June 9, 2015 No. 63 «On the Methodology for the Analysis, Optimization, Harmonization, and Description of Common Processes within the Eurasian Economic Union»;
Decision of the Board of the Eurasian Economic Commission of September 28, 2015 No. 125 «On Approval of the Regulation on the Exchange of Electronic Documents during Transboundary Interaction of Public Authorities of the Member States of the Eurasian Economic Union Among Themselves and with the Eurasian Economic Commission»;
Decision of the Board of the Eurasian Economic Commission of December 22, 2015 No. 174 «On Approval of the Rules for Conducting Monitoring of the Safety, Quality and Efficacy of Medical Devices».
II. Scope of Application
2. These Rules are developed for the purpose of determining the procedure and conditions for information interaction between the participants of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices» (hereinafter - the common process), including the description of the procedures performed within the framework of this common process.
3. These Rules are applied by the participants of the common process when monitoring the procedure for performing procedures and operations within the framework of the common process, as well as during the design, development, and refinement of information system components that ensure the implementation of the common process.
III. Basic Concepts
4. For the purposes of these Rules, concepts are used that mean the following:
«authorization» - granting a specific participant of the common process the rights to perform specific actions;
«unified database» - the unified information database for monitoring the safety, quality, and efficacy of medical devices, the formation and maintenance of which is carried out in electronic form by the Eurasian Economic Commission.
The concepts «group of common process procedures», «information object of the common process», «executor», «common process operation», «common process procedure», and «common process participant» used in these Rules are applied in the meanings defined by the Methodology for the Analysis, Optimization, Harmonization, and Description of Common Processes within the Eurasian Economic Union, approved by Decision of the Board of the Eurasian Economic Commission of June 9, 2015 No. 63.
The concept of «circulation of medical devices» used in these Rules is applied in the meaning defined by Article 2 of the Agreement on Common Principles and Rules for Circulation of Medical Devices (Medical Equipment and Medical Materials) within the Eurasian Economic Union of December 23, 2014.
IV. General Information About the Common Process
5. Full name of the common process: «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices».
6. Code designation of the common process: P.MM.08, version 1.0.0.
1. Goal and Objectives of the Common Process
7. The goals of the common process are:
a) creation of prerequisites for reducing costs associated with the exchange of information on the results of monitoring the safety, quality, and efficacy of medical devices, by creating a common information space in the field of circulation of medical devices;
b) creation of conditions for preventing the circulation within the territory of the Union of medical devices that do not meet the requirements of safety, quality, and efficacy, by promptly providing the participants of the common process with up-to-date information on medical devices by means of the integrated information system of foreign and mutual trade (hereinafter - the integrated system).
8. To achieve the goals of the common process, the following objectives must be met:
a) to create within the integrated system an information resource containing data on the results of monitoring the safety, quality, and efficacy of medical devices;
b) to ensure the possibility for the competent authorities of the Member States of the Union (hereinafter - the Member State) to obtain data from the unified database in electronic form and in an automated mode by means of the integrated system;
c) to ensure the possibility for interested parties and employees of the Eurasian Economic Commission (hereinafter - the Commission) to obtain up-to-date, complete, and reliable data from the unified database through the use of the information portal of the Union;
d) to ensure the use of unified classifiers and reference books in the formation and maintenance of the unified database.
2. Participants of the Common Process
9. The list of common process participants is given in Table 1.
Table 1
List of common process participants
| Code designation | Name | Description |
|---|---|---|
| P.ACT.001 | Commission | the body of the Union responsible for the formation and maintenance of the unified database, as well as the use of data from it |
| P.MM.08.ACT.001 | competent authority of the Member State | the competent authority of the Member State that submits data to the Commission to update the unified database and requests data from the unified database via the integrated system |
| P.MM.08.ACT.002 | interested party | a person obtaining data from the unified database through the use of the information portal of the Union |
3. Structure of the Common Process
10. The common process is a set of procedures grouped by their purpose:
a) procedures for the formation and maintenance of the unified database;
b) procedures for the provision of data from the unified database.
11. During the execution of the common process procedures, the competent authority of the Member State submits to the Commission data on the results of monitoring the safety, quality, and efficacy of medical devices. Based on the received data, the Commission makes changes to the unified database and publishes it on the information portal of the Union. The competent authority of the Member State requests up-to-date data from the unified database from the Commission.
Data on adverse events (incidents), corrective actions, results of investigations of an adverse event (incident), and post-registration clinical monitoring of the safety of medical devices are not subject to publication on the information portal of the Union.
Information interaction between the competent authority of the Member State and the Commission is carried out using the integrated system. Access of interested parties to the information contained in the unified database is carried out through the use of the information portal of the Union.
The provision of the specified data is carried out in accordance with the Regulation on Information Interaction between the competent authorities of the Member States of the Eurasian Economic Union and the Eurasian Economic Commission in the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices», approved by Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94 (hereinafter - the Regulation on Information Interaction).
The format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data used for the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices», approved by Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94 (hereinafter - the Description of Formats and Structures of Electronic Documents and Data).
12. The given description of the structure of the common process is presented in Figure 1.
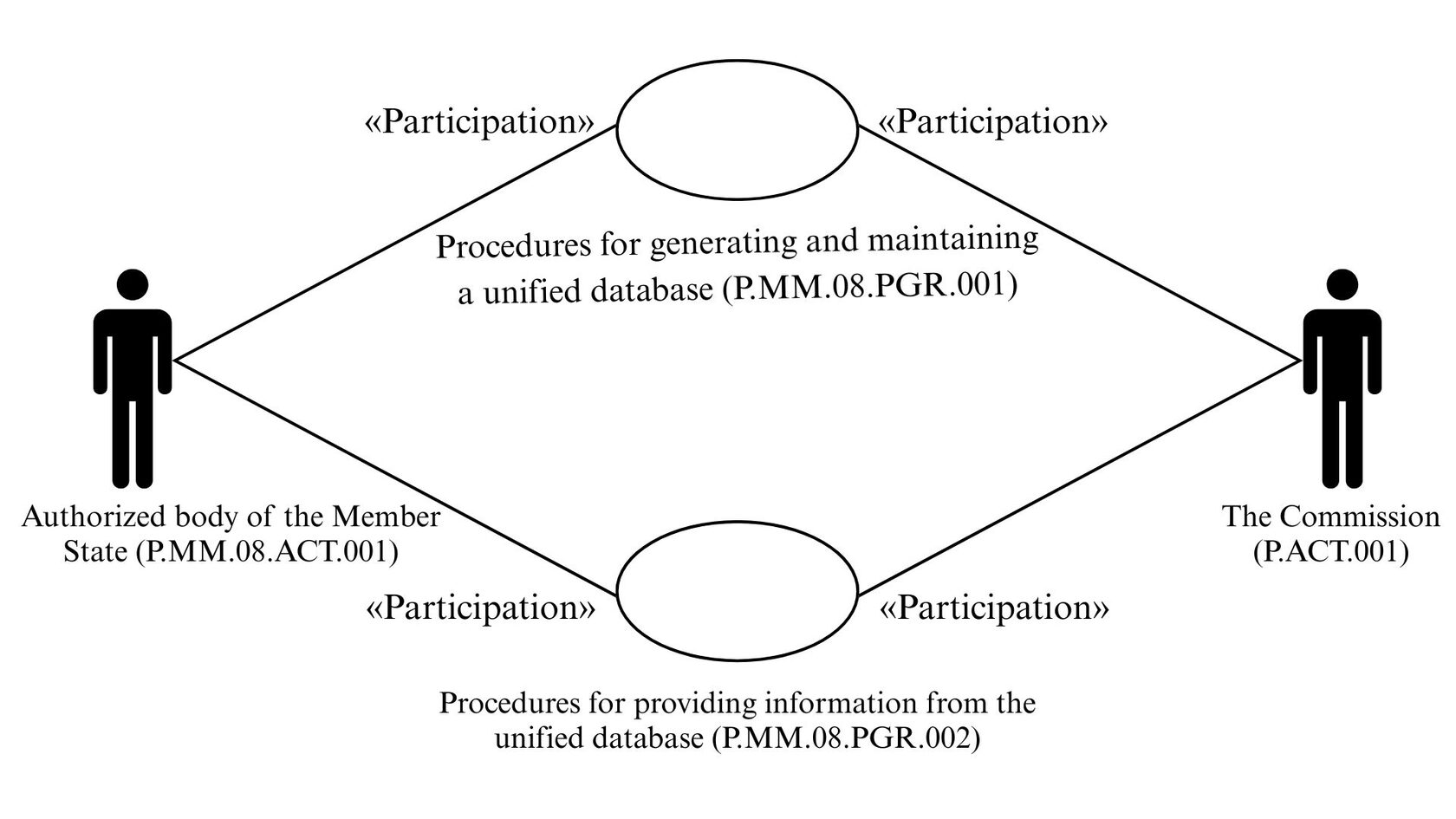
Fig. 1. Structure of the common process
13. The procedure for performing the common process procedures, grouped by their purpose, including a detailed description of the operations, is given in Section VIII of these Rules.
14. For each group of procedures, a general schema is provided demonstrating the relationships between the common process procedures and the sequence of their execution. The general schema of procedures is built using the UML (Unified Modeling Language) graphic notation and is provided with a textual description.
4. Group of Procedures for the Formation and Maintenance of the Unified Database
15. Procedures are performed as data on the results of monitoring the safety, quality, and efficacy of medical devices are received by the competent authority of the Member State. The competent authority of the Member State forms and sends the received data on the results of monitoring the safety, quality, and efficacy of medical devices to the Commission. Depending on the type of changes, the following procedures included in the group of procedures for the formation and maintenance of the unified database are performed:
«Inclusion of data into the unified database» (P.MM.08.PRC.001);
«Modification of data in the unified database» (P.MM.08.PRC.002).
16. The given description of the group of procedures for the formation and maintenance of the unified database is presented in Figure 2.

Fig. 2. General schema of the group of procedures for the formation and maintenance of the unified database
17. The list of common process procedures included in the group of procedures for the formation and maintenance of the unified database is given in Table 2.
Table 2
List of common process procedures included in the group of procedures for the formation and maintenance of the unified database
| Code designation | Name | Description |
|---|---|---|
| P.MM.08.PRC.001 | inclusion of data into the unified database | intended for the inclusion of data into the unified database. The procedure includes the submission of data by the competent authority of the Member State to the Commission, reception and processing of data, publication of data on the information portal of the Union, sending a notification of the inclusion of data into the unified database |
| P.MM.08.PRC.002 | modification of data in the unified database | intended for the modification of data in the unified database. The procedure includes the submission of data to the Commission, reception and processing of data, publication of data on the information portal of the Union, sending a notification of the modification of data in the unified database |
5. Group of Procedures for the Provision of Data from the Unified Database
18. The procedures for the provision of data from the unified database are carried out upon receiving requests from the information systems of the competent authorities of the Member States. At the same time, the following types of requests may be made:
request for information on the date and time of the unified database update;
request for data from the unified database;
request for modified data from the unified database.
The request for information on the date and time of the unified database update is carried out in order to synchronize the data stored in the information systems of the competent authorities of the Member States with the data contained in the unified database and stored at the Commission.
The request for data from the unified database is carried out to enable the competent authority of the Member State to obtain up-to-date data from the unified database for all Member States or for a specific Member State, depending on the conditions of the request. The request for data from the unified database can be made either as of the current date or the date specified in the request.
The request for modified data from the unified database is carried out to enable the competent authority of the Member State to obtain modified data on the results of monitoring the safety, quality, and efficacy of medical devices. Upon execution of the request, data from the unified database added or modified from the date and time specified in the request to the moment of request execution are provided. Depending on the conditions of the request, data from the unified database are provided for all Member States or for a specific Member State. This request is used during the initial data load, for example, upon initialization of the common process, upon connection of a new Member State, or data recovery after a failure.
Depending on the type of request, one of the following procedures included in the group of procedures for the provision of data from the unified database is performed:
«Obtaining information on the date and time of the unified database update» (P.MM.08.PRC.003);
«Obtaining data from the unified database» (P.MM.08.PRC.004);
«Obtaining modified data from the unified database» (P.MM.08.PRC.005).
These procedures can be performed sequentially, or each procedure can be performed separately from the others depending on the goals of the request.
19. The given description of the group of procedures for the provision of data from the unified database is presented in Figure 3.

Fig. 3. General schema of the group of procedures for the provision of data from the unified database
20. The list of common process procedures included in the group of procedures for the provision of data from the unified database is given in Table 3.
Table 3
List of common process procedures included in the group of procedures for the provision of data from the unified database
| Code designation | Name | Description |
|---|---|---|
| P.MM.08.PRC.003 | obtaining information on the date and time of the unified database update | intended for the provision of information on the date and time of the unified database update upon requests from the information systems of the competent authorities of the Member States through the integrated system |
| P.MM.08.PRC.004 | obtaining data from the unified database | intended for the provision of data from the unified database upon requests from the information systems of the competent authorities of the Member States through the integrated system |
| P.MM.08.PRC.005 | obtaining modified data from the unified database | intended for the provision of modified data from the unified database upon requests from the information systems of the competent authorities of the Member States through the integrated system |
V. Information Objects of the Common Process
21. The list of information objects, data about which or from which are transmitted in the process of interaction between the common process participants, is given in Table 4.
Table 4
List of information objects
| Code designation | Name | Description |
|---|---|---|
| P.MM.08.BEN.001 | unified database | unified information resource containing data on the results of monitoring the safety, quality, and efficacy of medical devices |
VI. Responsibility of the Common Process Participants
22. Bringing to disciplinary responsibility for non-compliance with the requirements aimed at ensuring the timeliness and completeness of data transmission of officials and employees of the Commission participating in information interaction is carried out in accordance with the Treaty on the Eurasian Economic Union of May 29, 2014, other international treaties and acts comprising the law of the Union, and of officials and employees of the competent authorities of the Member States - in accordance with the legislation of the Member States.
VII. Reference Books and Classifiers of the Common Process
23. The list of reference books and classifiers of the common process is given in Table 5.
Table 5
List of reference books and classifiers of the common process
| Code designation | Name | Type | Description |
|---|---|---|---|
| P.CLS.019 | classifier of countries of the world | classifier | contains a list of codes and names of countries of the world in accordance with the ISO 3166-1 standard |
| P.CLS.024 | language classifier | classifier | contains a list of language codes and names in accordance with the ISO 639-1 standard |
| P.CLS.048 | classifier of types of processing results for electronic documents and data | classifier | contains a list of codes and names for the types of processing results of electronic documents and data |
| P.CLS.054 | classifier of organizational and legal forms | classifier | contains a list of codes and names of organizational and legal forms |
| P.CLS.064 | nomenclature of medical devices of the Eurasian Economic Union | reference book | contains a list of types of medical devices specifying their nomenclature names, codes, and descriptions of the types of medical devices |
| P.MM.08.CLS.001 | classifier of types of adverse events | classifier | contains a list of codes and names of types of adverse events |
| P.MM.08.CLS.002 | classifier of user types of medical devices at the time of occurrence of adverse events (incidents) | classifier | contains a list of codes and names of user types of medical devices at the time of occurrence of adverse events (incidents) |
VIII. Common Process Procedures
1. Procedures for the formation and maintenance of the unified database
Procedure «Inclusion of data into the unified database» (P.MM.08.PRC.001)
24. The execution schema of the procedure «Inclusion of data into the unified database» (P.MM.08.PRC.001) is presented in Figure 4.

Fig. 4. Execution schema of the procedure «Inclusion of data into the unified database» (P.MM.08.PRC.001)
25. The procedure «Inclusion of data into the unified database» (P.MM.08.PRC.001) is executed in the event the competent authority of the Member State receives data on the results of monitoring the safety, quality, and efficacy of medical devices.
26. The first to be executed is the operation «Submission of data for inclusion into the unified database» (P.MM.08.OPR.001), following the results of which the competent authority of the Member State forms and sends to The Commission the data obtained as a result of monitoring the safety, quality, and efficacy of medical devices, for inclusion into the unified database.
27. Upon receipt by The Commission of data for inclusion into the unified database, the operation «Reception and processing of data for inclusion into the unified database» (P.MM.08.OPR.002) is executed, following the results of which The Commission receives the specified data.
28. Upon receipt by the competent authority of the Member State of the notification on the results of processing the data for inclusion into the unified database, the operation «Receipt of notification on the results of processing data for inclusion into the unified database» (P.MM.08.OPR.003) is executed, following the results of which the competent authority of the Member State that sent the data receives the notification on the results of the data processing.
29. In case of execution of the operation «Reception and processing of data for inclusion into the unified database» (P.MM.08.OPR.002), the operation «Publication of the unified database after data inclusion» (P.MM.08.OPR.004) is executed, following the results of which the updated unified database is published on the information portal of the Union.
30. The result of the execution of the procedure «Inclusion of data into the unified database» (P.MM.08.PRC.001) is the inclusion into the unified database of data obtained as a result of monitoring the safety, quality, and efficacy of medical devices, and the publication of the unified database on the information portal of the Union.
31. The list of common process operations performed within the framework of the procedure «Inclusion of data into the unified database» (P.MM.08.PRC.001) is given in Table 6.
Table 6
List of common process operations performed within the framework of the procedure «Inclusion of data into the unified database» (P.MM.08.PRC.001)
| Code designation | Name | Description |
|---|---|---|
| P.MM.08.OPR.001 | submission of data for inclusion into the unified database | given in Table 7 of these Rules |
| P.MM.08.OPR.002 | reception and processing of data for inclusion into the unified database | given in Table 8 of these Rules |
| P.MM.08.OPR.003 | receipt of notification on the results of processing data for inclusion into the unified database | given in Table 9 of these Rules |
| P.MM.08.OPR.004 | publication of the unified database after data inclusion | given in Table 10 of these Rules |
Table 7
Description of the operation «Submission of data for inclusion into the unified database» (P.MM.08.OPR.001)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.001 |
| 2 | Name of the operation | submission of data for inclusion into the unified database |
| 3 | Executor | competent authority of the Member State |
| 4 | Execution conditions | executed upon the inclusion of data obtained as a result of monitoring the safety, quality, and efficacy of medical devices into the unified database |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor forms the data for inclusion into the unified database and sends them to The Commission in accordance with the Regulation on Information Interaction |
| 7 | Results | data for inclusion into the unified database are transferred to The Commission |
Table 8
Description of the operation «Reception and processing of data for inclusion into the unified database» (P.MM.08.OPR.002)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.002 |
| 2 | Name of the operation | reception and processing of data for inclusion into the unified database |
| 3 | Executor | The Commission |
| 4 | Execution conditions | executed upon receipt of data for inclusion into the unified database (operation «Submission of data for inclusion into the unified database» (P.MM.08.OPR.001)) |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor receives the data for inclusion into the unified database and checks them in accordance with the Regulation on Information Interaction. In case of successful verification, the executor performs the inclusion of the data into the unified database specifying the date and time of data update and sends a notification to the competent authority of the Member State with the processing result code corresponding to the data inclusion, in accordance with the Regulation on Information Interaction |
| 7 | Results | the unified database is updated, the notification on the results of processing data for inclusion into the unified database is sent to the competent authority of the Member State |
Table 9
Description of the operation «Receipt of notification on the results of processing data for inclusion into the unified database» (P.MM.08.OPR.003)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.003 |
| 2 | Name of the operation | receipt of notification on the results of processing data for inclusion into the unified database |
| 3 | Executor | competent authority of the Member State |
| 4 | Execution conditions | executed upon receipt of the notification on the results of processing data for inclusion into the unified database (operation «Reception and processing of data for inclusion into the unified database» (P.MM.08.OPR.002)) |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor receives the notification on the results of processing data for inclusion into the unified database and checks it in accordance with the Regulation on Information Interaction |
| 7 | Results | notification on the results of processing data for inclusion into the unified database is received |
Table 10
Description of the operation «Publication of the unified database after data inclusion» (P.MM.08.OPR.004)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.004 |
| 2 | Name of the operation | publication of the unified database after data inclusion |
| 3 | Executor | The Commission |
| 4 | Execution conditions | executed after the unified database update (operation «Reception and processing of data for inclusion into the unified database» (P.MM.08.OPR.002)) |
| 5 | Constraints | data are published taking into account paragraph 11 of these Rules |
| 6 | Description of the operation | the executor publishes the unified database on the information portal of the Union |
| 7 | Results | updated unified database is published on the information portal of the Union |
Procedure «Modification of data in the unified database» (P.MM.08.PRC.002)
32. The execution schema of the procedure «Modification of data in the unified database» (P.MM.08.PRC.002) is presented in Figure 5.

Fig. 5. Execution schema of the procedure «Modification of data in the unified database» (P.MM.08.PRC.002)
33. The procedure «Modification of data in the unified database» (P.MM.08.PRC.002) is executed when it is necessary for the competent authority of the Member State to modify data obtained as a result of monitoring the safety, quality, and efficacy of medical devices.
34. The first to be executed is the operation «Submission of data to make modifications in the unified database» (P.MM.08.OPR.005), following the results of which the competent authority of the Member State forms and sends to The Commission the modified data obtained as a result of monitoring the safety, quality, and efficacy of medical devices, for updating the data in the unified database.
35. Upon receipt by The Commission of modified data obtained as a result of monitoring the safety, quality, and efficacy of medical devices, the operation «Reception and processing of data to make modifications in the unified database» (P.MM.08.OPR.006) is executed, following the results of which The Commission receives the specified data.
36. Upon receipt by the competent authority of the Member State of the notification on the processing results, the operation «Receipt of notification on the results of modifying data in the unified database» (P.MM.08.OPR.007) is executed, following the results of which the competent authority of the Member State that sent the data receives the notification on the results of modifying the data in the unified database.
37. After the execution of the operation «Reception and processing of data to make modifications in the unified database» (P.MM.08.OPR.006), the operation «Publication of the unified database after data modification» (P.MM.08.OPR.008) is executed, following the results of which the data obtained as a result of monitoring the safety, quality, and efficacy of medical devices are published on the information portal of the Union.
38. The result of the execution of the procedure «Modification of data in the unified database» (P.MM.08.PRC.002) is the publication of the updated unified database on the information portal of the Union. At the same time, only the data relevant as of the current date are published from the unified database.
39. The list of common process operations performed within the framework of the procedure «Modification of data in the unified database» (P.MM.08.PRC.002) is given in Table 11.
Table 11
List of common process operations performed within the framework of the procedure «Modification of data in the unified database» (P.MM.08.PRC.002)
| Code designation | Name | Description |
|---|---|---|
| P.MM.08.OPR.005 | submission of data to make modifications in the unified database | given in Table 12 of these Rules |
| P.MM.08.OPR.006 | reception and processing of data to make modifications in the unified database | given in Table 13 of these Rules |
| P.MM.08.OPR.007 | receipt of notification on the results of modifying data in the unified database | given in Table 14 of these Rules |
| P.MM.08.OPR.008 | publication of the unified database after data modification | given in Table 15 of these Rules |
Table 12
Description of the operation «Submission of data to make modifications in the unified database» (P.MM.08.OPR.005)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.005 |
| 2 | Name of the operation | submission of data to make modifications in the unified database |
| 3 | Executor | competent authority of the Member State |
| 4 | Execution conditions | executed upon modification of data obtained as a result of monitoring the safety, quality, and efficacy of medical devices |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor forms the modified data obtained as a result of monitoring the safety, quality, and efficacy of medical devices, and sends them to The Commission in accordance with the Regulation on Information Interaction |
| 7 | Results | modified data obtained as a result of monitoring the safety, quality, and efficacy of medical devices are submitted to The Commission |
Table 13
Description of the operation «Reception and processing of data to make modifications in the unified database» (P.MM.08.OPR.006)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.006 |
| 2 | Name of the operation | reception and processing of data to make modifications in the unified database |
| 3 | Executor | The Commission |
| 4 | Execution conditions | executed upon receipt of modified data obtained as a result of monitoring the safety, quality, and efficacy of medical devices (operation «Submission of data to make modifications in the unified database» (P.MM.08.OPR.005)) |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor receives the data and checks them in accordance with the Regulation on Information Interaction. In case of successful verification, the executor fills in the date and time of the end of validity of the data being modified in the unified database with the value of the date and time of the start of validity of the received actual data. The data being modified in the unified database are saved to ensure the possibility of viewing the history of changes and become unavailable for further processing. The executor adds the received actual data to the unified database, records the date and time of the data update, forms and sends a notification to the competent authority of the Member State about the successful update of the unified database, specifying the processing result code corresponding to the data modification, in accordance with the Regulation on Information Interaction |
| 7 | Results | the unified database is updated, a notification on the update of the unified database is sent to the competent authority of the Member State |
Table 14
Description of the operation «Receipt of notification on the results of modifying data in the unified database» (P.MM.08.OPR.007)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.007 |
| 2 | Name of the operation | receipt of notification on the results of modifying data in the unified database |
| 3 | Executor | competent authority of the Member State |
| 4 | Execution conditions | executed after the reception of data to make modifications in the unified database (operation «Reception and processing of data to make modifications in the unified database» (P.MM.08.OPR.006)) |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor receives the notification and checks it in accordance with the Regulation on Information Interaction |
| 7 | Results | notification on the results of modifying data in the unified database is received |
Table 15
Description of the operation «Publication of the unified database after data modification» (P.MM.08.OPR.008)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.008 |
| 2 | Name of the operation | publication of the unified database after data modification |
| 3 | Executor | The Commission |
| 4 | Execution conditions | executed after making modifications in the unified database (operation «Reception and processing of data to make modifications in the unified database» (P.MM.08.OPR.006)) |
| 5 | Constraints | data are published in the volume specified in paragraph 11 of these Rules |
| 6 | Description of the operation | the executor publishes the modified data obtained as a result of monitoring the safety, quality, and efficacy of medical devices on the information portal of the Union |
| 7 | Results | updated unified database is published on the information portal of the Union |
2. Procedures for the provision of data from the unified database
Procedure «Obtaining information on the date and time of the unified database update» (P.MM.08.PRC.003)
40. The execution schema of the procedure «Obtaining information on the date and time of the unified database update» (P.MM.08.PRC.003) is presented in Figure 6.

Fig. 6. Execution schema of the procedure «Obtaining information on the date and time of the unified database update» (P.MM.08.PRC.003)
41. The procedure «Obtaining information on the date and time of the unified database update» (P.MM.08.PRC.003) is executed by the competent authority of the Member State when it is necessary to obtain information on the date and time of the unified database update.
42. The first to be executed is the operation «Request for information on the date and time of the unified database update» (P.MM.08.OPR.009), following the results of which the competent authority of the Member State forms and sends to The Commission a request to provide information on the date and time of the unified database update.
43. Upon receipt by The Commission of the request to provide information on the date and time of the unified database update, the operation «Processing and provision of information on the date and time of the unified database update» (P.MM.08.OPR.010) is executed, following the results of which The Commission sends to the competent authority of the Member State the information on the date and time of the unified database update.
44. Upon receipt by the competent authority of the Member State of the information on the date and time of the unified database update, the operation «Reception and processing of information on the date and time of the unified database update» (P.MM.08.OPR.011) is executed, following the results of which the competent authority of the Member State that sent the request to provide information on the date and time of the unified database update, processes the received information on the date and time of the unified database update.
45. The result of the execution of the procedure «Obtaining information on the date and time of the unified database update» (P.MM.08.PRC.003) is the receipt by the competent authority of the Member State of the information on the date and time of the unified database update.
46. The list of common process operations performed within the framework of the procedure «Obtaining information on the date and time of the unified database update» (P.MM.08.PRC.003) is given in Table 16.
Table 16
List of common process operations performed within the framework of the procedure «Obtaining information on the date and time of the unified database update» (P.MM.08.PRC.003)
| Code designation | Name | Description |
|---|---|---|
| P.MM.08.OPR.009 | request for information on the date and time of the unified database update | given in Table 17 of these Rules |
| P.MM.08.OPR.010 | processing and provision of information on the date and time of the unified database update | given in Table 18 of these Rules |
| P.MM.08.OPR.011 | reception and processing of information on the date and time of the unified database update | given in Table 19 of these Rules |
Table 17
Description of the operation «Request for information on the date and time of the unified database update» (P.MM.08.OPR.009)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.009 |
| 2 | Name of the operation | request for information on the date and time of the unified database update |
| 3 | Executor | competent authority of the Member State |
| 4 | Execution conditions | executed when it is necessary to obtain information on the date and time of the unified database update |
| 5 | Constraints | the format and structure of the request must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor sends to The Commission a request to provide information on the date and time of the unified database update in accordance with the Regulation on Information Interaction |
| 7 | Results | request to provide information on the date and time of the unified database update is sent |
Table 18
Description of the operation «Processing and provision of information on the date and time of the unified database update» (P.MM.08.OPR.010)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.010 |
| 2 | Name of the operation | processing and provision of information on the date and time of the unified database update |
| 3 | Executor | The Commission |
| 4 | Execution conditions | executed upon receipt of the request to provide information on the date and time of the unified database update (operation «Request for information on the date and time of the unified database update» (P.MM.08.OPR.009)) |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor verifies the received request and sends a response to the request in accordance with the Regulation on Information Interaction |
| 7 | Results | information on the date and time of the unified database update is provided to the competent authority of the Member State |
Table 19
Description of the operation «Reception and processing of information on the date and time of the unified database update» (P.MM.08.OPR.011)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.011 |
| 2 | Name of the operation | reception and processing of information on the date and time of the unified database update |
| 3 | Executor | competent authority of the Member State |
| 4 | Execution conditions | executed upon receiving information on the date and time of the unified database update (operation «Processing and provision of information on the date and time of the unified database update» (P.MM.08.OPR.010)) |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor checks the received information on the date and time of the unified database update in accordance with the Regulation on Information Interaction |
| 7 | Results | information on the date and time of the unified database update is received |
Procedure «Obtaining data from the unified database» (P.MM.08.PRC.004)
47. The execution schema of the procedure «Obtaining data from the unified database» (P.MM.08.PRC.004) is presented in Figure 7.

Fig. 7. Execution schema of the procedure «Obtaining data from the unified database» (P.MM.08.PRC.004)
48. The procedure «Obtaining data from the unified database» (P.MM.08.PRC.004) is executed by the competent authority of the Member State when it is necessary to obtain data from the unified database.
49. The first to be executed is the operation «Request for data from the unified database» (P.MM.08.OPR.012), following the results of which the competent authority of the Member State forms and sends to The Commission a request to provide data from the unified database.
50. Upon receipt by The Commission of the request to provide data from the unified database, the operation «Processing and provision of data from the unified database» (P.MM.08.OPR.013) is executed, following the results of which The Commission forms and sends to the competent authority of the Member State the data from the unified database or a notification on the absence of data satisfying the request parameters.
51. Upon receipt by the competent authority of the Member State of data from the unified database, the operation «Reception and processing of data from the unified database» (P.MM.08.OPR.014) is executed, following the results of which the competent authority of the Member State that sent the request to provide data from the unified database processes the received data from the unified database or the notification on the absence of data satisfying the request parameters.
52. The result of the execution of the procedure «Obtaining data from the unified database» (P.MM.08.PRC.004) is the receipt by the competent authority of the Member State of data from the unified database or a notification on the absence of data satisfying the request parameters.
53. The list of common process operations performed within the framework of the procedure «Obtaining data from the unified database» (P.MM.08.PRC.004) is given in Table 20.
Table 20
List of common process operations performed within the framework of the procedure «Obtaining data from the unified database» (P.MM.08.PRC.004)
| Code designation | Name | Description |
|---|---|---|
| P.MM.08.OPR.012 | request for data from the unified database | given in Table 21 of these Rules |
| P.MM.08.OPR.013 | processing and provision of data from the unified database | given in Table 22 of these Rules |
| P.MM.08.OPR.014 | reception and processing of data from the unified database | given in Table 23 of these Rules |
Table 21
Description of the operation «Request for data from the unified database» (P.MM.08.OPR.012)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.012 |
| 2 | Name of the operation | request for data from the unified database |
| 3 | Executor | competent authority of the Member State |
| 4 | Execution conditions | executed when it is necessary for the competent authority of the Member State to obtain data from the unified database |
| 5 | Constraints | the format and structure of the request must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor sends to The Commission a request to provide data from the unified database in accordance with the Regulation on Information Interaction. The executor requests actual data for all Member States or for a specific Member State, specifying its code in the request, with the indication of the date and time for which the actual data are provided. If the date is not specified, all actual data contained in the unified database as of the current date are provided |
| 7 | Results | request to obtain data from the unified database is sent |
Table 22
Description of the operation «Processing and provision of data from the unified database» (P.MM.08.OPR.013)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.013 |
| 2 | Name of the operation | processing and provision of data from the unified database |
| 3 | Executor | The Commission |
| 4 | Execution conditions | executed upon receipt of the request to provide data from the unified database (operation «Request for data from the unified database» (P.MM.08.OPR.012)) |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor verifies the received request in accordance with the Regulation on Information Interaction, forms and sends to the competent authority of the Member State the data from the unified database or a notification on the absence of data satisfying the request parameters, specifying the processing result code corresponding to the absence of data. Data from the unified database are provided as of the date specified in the request (data for which the start date is less than the one specified in the request, and the end date is greater than the one specified in the request or is not set). In case the country code is specified in the request, the response message provides data from the unified database for the specified Member State; if the country code is not specified - for all Member States |
| 7 | Results | data from the unified database are provided to the competent authority of the Member State or a notification on the absence of data satisfying the request parameters is sent |
Table 23
Description of the operation «Reception and processing of data from the unified database» (P.MM.08.OPR.014)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.014 |
| 2 | Name of the operation | reception and processing of data from the unified database |
| 3 | Executor | competent authority of the Member State |
| 4 | Execution conditions | executed upon provision of data from the unified database or a notification on the absence of data satisfying the request parameters (operation «Processing and provision of data from the unified database» (P.MM.08.OPR.013)) |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor checks the received data in accordance with the Regulation on Information Interaction |
| 7 | Results | data from the unified database or a notification on the absence of data satisfying the request parameters are received |
Procedure «Obtaining modified data from the unified database» (P.MM.08.PRC.005)
54. The execution schema of the procedure «Obtaining modified data from the unified database» (P.MM.08.PRC.005) is presented in Figure 8.

Fig. 8. Execution schema of the procedure «Obtaining modified data from the unified database» (P.MM.08.PRC.005)
55. The procedure «Obtaining modified data from the unified database» (P.MM.08.PRC.005) is executed by the competent authority of the Member State when it is necessary to obtain modified data from the unified database, the inclusion or modification of which occurred starting from the moment specified in the request until the moment of execution of this request. The procedure is executed, inter alia, if as a result of executing the procedure «Obtaining information on the date and time of the unified database update» (P.MM.08.PRC.003), it is revealed that the date and time of the last receipt of data from the unified database by the competent authority of the Member State are earlier than the date and time of the last update of the unified database.
56. The first to be executed is the operation «Request for modified data from the unified database» (P.MM.08.OPR.015), following the results of which the competent authority of the Member State forms and sends to The Commission a request to provide modified data from the unified database.
57. Upon receipt by The Commission of the request to provide modified data from the unified database, the operation «Processing and provision of modified data from the unified database» (P.MM.08.OPR.016) is executed, following the results of which The Commission forms and sends to the competent authority of the Member State the data modified in the unified database starting from the date specified in the request, or a notification on the absence of data satisfying the request parameters.
58. Upon receipt by the competent authority of the Member State of the data modified in the unified database or the notification on the absence of data satisfying the request parameters, the operation «Reception and processing of modified data from the unified database» (P.MM.08.OPR.017) is executed, following the results of which the competent authority of the Member State that sent the request to provide data modified in the unified database processes the received data or the notification on the absence of data satisfying the request parameters.
59. The result of the execution of the procedure «Obtaining modified data from the unified database» (P.MM.08.PRC.005) is the receipt by the competent authority of the Member State of modified data from the unified database or a notification on the absence of data satisfying the request parameters.
60. The list of common process operations performed within the framework of the procedure «Obtaining modified data from the unified database» (P.MM.08.PRC.005) is given in Table 24.
Table 24
List of common process operations performed within the framework of the procedure «Obtaining modified data from the unified database» (P.MM.08.PRC.005)
| Code designation | Name | Description |
|---|---|---|
| P.MM.08.OPR.015 | request for modified data from the unified database | given in Table 25 of these Rules |
| P.MM.08.OPR.016 | processing and provision of modified data from the unified database | given in Table 26 of these Rules |
| P.MM.08.OPR.017 | reception and processing of modified data from the unified database | given in Table 27 of these Rules |
Table 25
Description of the operation «Request for modified data from the unified database» (P.MM.08.OPR.015)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.015 |
| 2 | Name of the operation | request for modified data from the unified database |
| 3 | Executor | competent authority of the Member State |
| 4 | Execution conditions | executed when it is necessary to obtain modified data from the unified database |
| 5 | Constraints | the format and structure of the request must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor sends to The Commission a request to provide modified data from the unified database starting from the date and time of update specified in the request until the moment of execution of the request, in accordance with the Regulation on Information Interaction. To request modified data from the unified database in full, the date in the request is not filled in. If it is necessary to request the provision of modified data for a specific Member State, its code must be specified in the request. If the country code is not specified in the request, modified data for all Member States are provided |
| 7 | Results | request to provide modified data from the unified database is sent |
Table 26
Description of the operation «Processing and provision of modified data from the unified database» (P.MM.08.OPR.016)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.016 |
| 2 | Name of the operation | processing and provision of modified data from the unified database |
| 3 | Executor | The Commission |
| 4 | Execution conditions | executed upon receipt of the request to provide modified data from the unified database (operation «Request for modified data from the unified database» (P.MM.08.OPR.015)) |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor verifies the received request in accordance with the Regulation on Information Interaction, forms and sends to the competent authority of the Member State a message with modified data from the unified database starting from the date and time of update specified in the request, or a notification on the absence of data satisfying the request parameters, specifying the processing result code corresponding to the absence of data. Modified data from the unified database are provided for all Member States or for a specific Member State depending on the conditions of the request. Modified data from the unified database are provided taking into account the history of changes |
| 7 | Results | modified data from the unified database are provided to the competent authority of the Member State or a notification on the absence of data satisfying the request parameters is sent |
Table 27
Description of the operation «Reception and processing of modified data from the unified database» (P.MM.08.OPR.017)
| No. | Element designation | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.OPR.017 |
| 2 | Name of the operation | reception and processing of modified data from the unified database |
| 3 | Executor | competent authority of the Member State |
| 4 | Execution conditions | executed upon provision of modified data from the unified database or a notification on the absence of data satisfying the request parameters (operation «Processing and provision of modified data from the unified database» (P.MM.08.OPR.016)) |
| 5 | Constraints | the format and structure of the submitted data must comply with the Description of Formats and Structures of Electronic Documents and Data |
| 6 | Description of the operation | the executor checks the received data in accordance with the Regulation on Information Interaction |
| 7 | Results | modified data from the unified database or a notification on the absence of modified data satisfying the request parameters are received |
IX. Procedure for actions in non-standard situations
61. When performing common process procedures, exceptional situations are possible in which data processing cannot be carried out in a normal mode. This can happen in the event of technical failures, errors in structural and format-logical control, and in other cases.
62. In the event of errors in structural and format-logical control, the competent authority of the Member State checks the message regarding which an error notification was received for compliance with the Description of Formats and Structures of Electronic Documents and Data and the requirements for filling out electronic documents and data in accordance with the Regulation on Information Interaction. If a non-compliance of the data with the requirements of the specified documents is detected, the competent authority of the Member State takes the necessary measures to eliminate the detected error in the prescribed manner.
63. In order to resolve non-standard situations, the Member States inform each other and The Commission about the competent authorities of the Member States whose competence includes fulfilling the requirements provided for by these Rules, and also provide information on the persons responsible for providing technical support during the implementation of the common process.
Approved
by Decision of the Board
of the Eurasian Economic Commission
of August 30, 2016 No. 94
REGULATION
ON INFORMATION INTERACTION BETWEEN THE COMPETENT AUTHORITIES OF THE MEMBER STATES OF THE EURASIAN ECONOMIC UNION AND THE EURASIAN ECONOMIC COMMISSION IN THE IMPLEMENTATION, BY MEANS OF THE INTEGRATED INFORMATION SYSTEM OF FOREIGN AND MUTUAL TRADE, OF THE COMMON PROCESS «FORMATION, MAINTENANCE AND USE OF THE UNIFIED INFORMATION DATABASE FOR MONITORING THE SAFETY, QUALITY AND EFFICACY OF MEDICAL DEVICES»
I. General Provisions
1. This Regulation is developed in accordance with the following acts comprising the law of the Eurasian Economic Union (hereinafter - the Union):
Treaty on the Eurasian Economic Union of May 29, 2014;
Agreement on Common Principles and Rules for Circulation of Medical Devices (Medical Equipment and Medical Materials) within the Eurasian Economic Union of December 23, 2014;
Decision of the Supreme Eurasian Economic Council of December 23, 2014 No. 109 «On the Implementation of the Agreement on Common Principles and Rules for Circulation of Medical Devices (Medical Equipment and Medical Materials) within the Eurasian Economic Union»;
Decision of the Council of the Eurasian Economic Commission of February 12, 2016 No. 30 «On Approval of the Procedure for the Formation and Maintenance of the Information System in the Field of Circulation of Medical Devices»;
Decision of the Board of the Eurasian Economic Commission of November 6, 2014 No. 200 «On Technological Documents Regulating Information Interaction in the Implementation of Common Processes by Means of the Integrated Information System of Foreign and Mutual Trade»;
Decision of the Board of the Eurasian Economic Commission of January 27, 2015 No. 5 «On Approval of the Rules for Electronic Data Exchange in the Integrated Information System of Foreign and Mutual Trade»;
Decision of the Board of the Eurasian Economic Commission of April 14, 2015 No. 29 «On the List of Common Processes within the Eurasian Economic Union and the Introduction of an Amendment to Decision of the Board of the Eurasian Economic Commission of August 19, 2014 No. 132»;
Decision of the Board of the Eurasian Economic Commission of June 9, 2015 No. 63 «On the Methodology for the Analysis, Optimization, Harmonization, and Description of Common Processes within the Eurasian Economic Union»;
Decision of the Board of the Eurasian Economic Commission of September 28, 2015 No. 125 «On Approval of the Regulation on the Exchange of Electronic Documents during Transboundary Interaction of Public Authorities of the Member States of the Eurasian Economic Union Among Themselves and with the Eurasian Economic Commission»;
Decision of the Board of the Eurasian Economic Commission of December 22, 2015 No. 174 «On Approval of the Rules for Conducting Monitoring of the Safety, Quality and Efficacy of Medical Devices».
II. Scope of Application
2. This Regulation is developed in order to ensure a uniform understanding by the participants of the common process of the procedure and conditions for executing the transactions of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices» (hereinafter - the common process), as well as their role during their execution.
3. This Regulation determines the requirements for the procedure and conditions of executing the operations of the common process directly aimed at implementing information interaction between the participants of the common process.
4. This Regulation is applied by the participants of the common process when monitoring the procedure for executing procedures and operations within the framework of the common process, as well as during the design, development, and refinement of components of information systems ensuring the implementation of this common process.
III. Basic Concepts
5. For the purposes of this Regulation, concepts are used that mean the following:
«attribute of an electronic document (data)» - a data element of an electronic document (data) that is considered indivisible in a specific context;
«state of an information object» - a property characterizing an information object at a certain stage of executing a common process procedure, which changes during the execution of common process operations.
The concepts of «initiator», «respondent», and «common process transaction» are used in this Regulation in the meanings defined by Decision of the Board of the Eurasian Economic Commission of June 9, 2015 No. 63 «On the Methodology for the Analysis, Optimization, Harmonization, and Description of Common Processes within the Eurasian Economic Union».
Other concepts used in this Regulation are applied in the meanings defined in paragraph 4 of the Rules for information interaction in the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices», approved by Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94 (hereinafter - the Rules for Information Interaction).
IV. General Information About Information Interaction within the Common Process
1. Participants of information interaction
6. The list of roles of participants in information interaction within the common process is given in Table 1.
Table 1
List of roles of information interaction participants
| Name of the role | Description of the role | Participant performing the role |
|---|---|---|
| Data owner | submits data to The Commission on the results of monitoring the safety, quality, and efficacy of medical devices to update the unified information database for monitoring the safety, quality, and efficacy of medical devices (hereinafter - the unified database) on the information portal of the Union; sends a request and receives data from the unified database through the integrated information system of foreign and mutual trade (hereinafter - the integrated system) | competent authority of the Member State (P.MM.08.ACT.001) |
| Coordinator | is responsible for the formation and maintenance of the unified database. Provides access to up-to-date data contained in the unified database | The Eurasian Economic Commission (P.ACT.001) |
2. Structure of information interaction
7. Information interaction within the common process is carried out between the competent authorities of the Member States of the Union and the Eurasian Economic Commission (hereinafter, respectively - the competent authority of the Member State, The Commission) in accordance with the common process procedures:
information interaction during the formation and maintenance of the unified database;
information interaction during the provision of data from the unified database.
The structure of information interaction between the competent authorities of the Member States and The Commission is presented in Figure 1.

Fig. 1. Structure of information interaction between the competent authorities of the Member States and The Commission
8. Information interaction between the competent authorities of the Member States and The Commission is implemented within the framework of the common process. The structure of the common process is defined in the Rules for Information Interaction.
9. Information interaction determines the procedure for executing common process transactions, each of which is an exchange of messages for the purpose of synchronizing the states of a common process information object among the participants of the common process. For each information interaction, the relationships between operations and the corresponding common process transactions are defined.
10. When executing a common process transaction, the initiator, within the framework of the operation being carried out by them (initiating operation), sends a request message to the respondent, in response to which the respondent, within the framework of the operation being carried out by them (receiving operation), may or may not send a response message depending on the template of the common process transaction. The structure of the data in the message must comply with the Description of Formats and Structures of Electronic Documents and Data used for the implementation of the common process by means of the integrated information system of foreign and mutual trade, approved by Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94 (hereinafter - the Description of Formats and Structures of Electronic Documents and Data).
11. Common process transactions are executed in accordance with the specified parameters of common process transactions, as defined by this Regulation.
V. Information Interaction within the Framework of Groups of Procedures
1. Information interaction during the formation and maintenance of the unified database
12. The schema for executing common process transactions during the formation and maintenance of the unified database is presented in Figure 2. For each common process procedure, Table 2 shows the relationship between operations, intermediate and resulting states of common process information objects, and common process transactions.

Fig. 2. Schema for executing common process transactions during the formation and maintenance of the unified database
Table 2
List of common process transactions during the formation and maintenance of the unified database
| No. | Operation performed by the initiator | Intermediate state of the information object of the common process | Operation performed by the respondent | Resulting state of the information object of the common process | Common process transaction |
|---|---|---|---|---|---|
| 1. Inclusion of data into the unified database (P.MM.08.PRC.001) | |||||
| 1.1 | Submission of data for inclusion into the unified database (P.MM.08.OPR.001). Receipt of notification on the results of processing data for inclusion into the unified database (P.MM.08.OPR.003) | unified database (P.MM.08.BEN.001): data for inclusion are transferred | reception and processing of data for inclusion into the unified database (P.MM.08.OPR.002) | unified database (P.MM.08.BEN.001): updated | transmission of data on the results of monitoring the safety of medical devices for inclusion into the unified database (P.MM.08.TRN.001) |
| 2. Modification of data in the unified database (P.MM.08.PRC.002) | |||||
| 2.1 | Submission of data to make modifications in the unified database (P.MM.08.OPR.005). Receipt of notification on the results of modifying data in the unified database (P.MM.08.OPR.007) | unified database (P.MM.08.BEN.001): data for modification are transferred | reception and processing of data to make modifications in the unified database (P.MM.08.OPR.006) | unified database (P.MM.08.BEN.001): updated | transmission of data on the results of monitoring the safety of medical devices for modification in the unified database (P.MM.08.TRN.002) |
2. Information interaction during the provision of data from the unified database
13. The schema for executing common process transactions during the provision of data from the unified database is presented in Figure 3. For each common process procedure, Table 3 shows the relationship between operations, intermediate and resulting states of common process information objects, and common process transactions.

Fig. 3. Schema for executing common process transactions during the provision of data from the unified database
Table 3
List of common process transactions during the provision of data from the unified database
| No. | Operation performed by the initiator | Intermediate state of the information object of the common process | Operation performed by the respondent | Resulting state of the information object of the common process | Common process transaction |
|---|---|---|---|---|---|
| 1. Obtaining information on the date and time of the unified database update (P.MM.08.PRC.003) | |||||
| 1.1 | Request for information on the date and time of the unified database update (P.MM.08.OPR.009). Reception and processing of information on the date and time of the unified database update (P.MM.08.OPR.011) | unified database (P.MM.08.BEN.001): information on the date and time of update is requested | processing and provision of information on the date and time of the unified database update (P.MM.08.OPR.010) | unified database (P.MM.08.BEN.001): information on the date and time of update is provided | obtaining information on the date and time of the unified database update (P.MM.08.TRN.003) |
| 2. Obtaining data from the unified database (P.MM.08.PRC.004) | |||||
| 2.1 | Request for data from the unified database (P.MM.08.OPR.012). Reception and processing of data from the unified database (P.MM.08.OPR.014) | unified database (P.MM.08.BEN.001): data are requested | processing and provision of data from the unified database (P.MM.08.OPR.013) | unified database (P.MM.08.BEN.001): data are provided. unified database (P.MM.08.BEN.001): data are absent | obtaining data from the unified database (P.MM.08.TRN.004) |
| 3. Obtaining modified data from the unified database (P.MM.08.PRC.005) | |||||
| 3.1 | Request for modified data from the unified database (P.MM.08.OPR.015). Reception and processing of modified data from the unified database (P.MM.08.OPR.017) | unified database (P.MM.08.BEN.001): modified data are requested | processing and provision of modified data from the unified database (P.MM.08.OPR.016) | unified database (P.MM.08.BEN.001): modified data are provided. unified database (P.MM.08.BEN.001): modified data are absent | obtaining modified data from the unified database (P.MM.08.TRN.005) |
VI. Description of Common Process Messages
14. The list of common process messages transmitted within the framework of information interaction during the implementation of the common process is given in Table 4. The structure of the data in the message must comply with the Description of Formats and Structures of Electronic Documents and Data. A link to the corresponding structure in the Description of Formats and Structures of Electronic Documents and Data is established by the value of column 3 in Table 4.
Table 4
List of common process messages
| Code designation | Name | Structure of the electronic document (data) |
|---|---|---|
| P.MM.08.MSG.001 | data on the results of monitoring the safety of medical devices for inclusion into the unified database | data on the results of monitoring the safety, quality, and efficacy of medical devices (R.HC.MM.08.001) |
| P.MM.08.MSG.002 | data on the results of monitoring the safety of medical devices for modification in the unified database | data on the results of monitoring the safety, quality, and efficacy of medical devices (R.HC.MM.08.001) |
| P.MM.08.MSG.003 | notification on the unified database update | notification on the processing result (R.006) |
| P.MM.08.MSG.004 | request for information on the date and time of the unified database update | state of updating of a shared resource (R.007) |
| P.MM.08.MSG.005 | information on the date and time of the unified database update | state of updating of a shared resource (R.007) |
| P.MM.08.MSG.006 | request for data from the unified database | state of updating of a shared resource (R.007) |
| P.MM.08.MSG.007 | data from the unified database | data on the results of monitoring the safety, quality, and efficacy of medical devices (R.HC.MM.08.001) |
| P.MM.08.MSG.008 | notification on the absence of data in the unified database | notification on the processing result (R.006) |
| P.MM.08.MSG.009 | request for modified data from the unified database | state of updating of a shared resource (R.007) |
| P.MM.08.MSG.010 | modified data from the unified database | data on the results of monitoring the safety, quality, and efficacy of medical devices (R.HC.MM.08.001) |
| P.MM.08.MSG.011 | notification on the absence of modified data in the unified database | notification on the processing result (R.006) |
VII. Description of Common Process Transactions
1. Common process transaction «Transmission of data on the results of monitoring the safety of medical devices for inclusion into the unified database» (P.MM.08.TRN.001)
15. The common process transaction «Transmission of data on the results of monitoring the safety of medical devices for inclusion into the unified database» (P.MM.08.TRN.001) is executed to transmit the corresponding data to the respondent. The execution schema of the specified common process transaction is presented in Figure 4. The parameters of the common process transaction are given in Table 5.

Fig. 4. Execution schema of the common process transaction «Transmission of data on the results of monitoring the safety of medical devices for inclusion into the unified database» (P.MM.08.TRN.001)
Table 5
Description of the common process transaction «Transmission of data on the results of monitoring the safety of medical devices for inclusion into the unified database» (P.MM.08.TRN.001)
| No. | Mandatory element | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.TRN.001 |
| 2 | Name of the common process transaction | transmission of data on the results of monitoring the safety of medical devices for inclusion into the unified database |
| 3 | Template of the common process transaction | request/response |
| 4 | Initiating role | initiator |
| 5 | Initiating operation | sending data for inclusion into the unified database |
| 6 | Responding role | respondent |
| 7 | Receiving operation | reception and processing of data for inclusion into the unified database |
| 8 | Result of executing the common process transaction | unified database (P.MM.08.BEN.001): updated |
| 9 | Parameters of the common process transaction: - time to confirm receipt - time to confirm acceptance for processing - response waiting time - authorization flag - number of retries | - 20 min 1 h yes 3 |
| 10 | Messages of the common process transaction: initiating message response message | data on the results of monitoring the safety of medical devices for inclusion into the unified database (P.MM.08.MSG.001) notification on the unified database update (P.MM.08.MSG.003) |
| 11 | Parameters of common process transaction messages: - EDS flag - transmission of an electronic document with an incorrect EDS | no - for P.MM.08.MSG.001 (except for cases when the use of an EDS when carrying out information interaction within the common process is provided for by the corresponding decision of the Board of The Commission) no - for P.MM.08.MSG.003 - |
2. Common process transaction «Transmission of data on the results of monitoring the safety of medical devices for modification in the unified database» (P.MM.08.TRN.002)
16. The common process transaction «Transmission of data on the results of monitoring the safety of medical devices for modification in the unified database» (P.MM.08.TRN.002) is executed to transmit the corresponding data to the respondent. The execution schema of the specified common process transaction is presented in Figure 5. The parameters of the common process transaction are given in Table 6.

Fig. 5. Execution schema of the common process transaction «Transmission of data on the results of monitoring the safety of medical devices for modification in the unified database» (P.MM.08.TRN.002)
Table 6
Description of the common process transaction «Transmission of data on the results of monitoring the safety of medical devices for modification in the unified database» (P.MM.08.TRN.002)
| No. | Mandatory element | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.TRN.002 |
| 2 | Name of the common process transaction | transmission of data on the results of monitoring the safety of medical devices for modification in the unified database |
| 3 | Template of the common process transaction | request/response |
| 4 | Initiating role | initiator |
| 5 | Initiating operation | sending data for modification in the unified database |
| 6 | Responding role | respondent |
| 7 | Receiving operation | reception and processing of data to make modifications in the unified database |
| 8 | Result of executing the common process transaction | unified database (P.MM.08.BEN.001): updated |
| 9 | Parameters of the common process transaction: - time to confirm receipt - time to confirm acceptance for processing - response waiting time - authorization flag - number of retries | - 20 min 1 h yes 3 |
| 10 | Messages of the common process transaction: initiating message response message | data on the results of monitoring the safety of medical devices for inclusion into the unified database (P.MM.08.MSG.002) notification on the unified database update (P.MM.08.MSG.003) |
| 11 | Parameters of common process transaction messages: - EDS flag - transmission of an electronic document with an incorrect EDS | no - for P.MM.08.MSG.002 (except for cases when the use of an EDS when carrying out information interaction within the common process is provided for by the corresponding decision of the Board of The Commission) no - for P.MM.08.MSG.003 - |
3. Common process transaction «Obtaining information on the date and time of the unified database update» (P.MM.08.TRN.003)
17. The common process transaction «Obtaining information on the date and time of the unified database update» (P.MM.08.TRN.003) is executed to provide corresponding data by the respondent upon the request of the initiator. The execution schema of the specified common process transaction is presented in Figure 6. The parameters of the common process transaction are given in Table 7.

Fig. 6. Execution schema of the common process transaction «Obtaining information on the date and time of the unified database update» (P.MM.08.TRN.003)
Table 7
Description of the common process transaction «Obtaining information on the date and time of the unified database update» (P.MM.08.TRN.003)
| No. | Mandatory element | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.TRN.003 |
| 2 | Name of the common process transaction | obtaining information on the date and time of the unified database update |
| 3 | Template of the common process transaction | query/response |
| 4 | Initiating role | initiator |
| 5 | Initiating operation | request and receipt of information on the date and time of the unified database update |
| 6 | Responding role | respondent |
| 7 | Receiving operation | provision of information on the date and time of the unified database update |
| 8 | Result of executing the common process transaction | unified database (P.MM.08.BEN.001): information on the date and time of update is provided |
| 9 | Parameters of the common process transaction: - time to confirm receipt - time to confirm acceptance for processing - response waiting time - authorization flag - number of retries | - - 1 h yes 3 |
| 10 | Messages of the common process transaction: initiating message response message | request for information on the date and time of the unified database update (P.MM.08.MSG.004) information on the date and time of the unified database update (P.MM.08.MSG.005) |
| 11 | Parameters of common process transaction messages: - EDS flag - transmission of an electronic document with an incorrect EDS | no - |
4. Common process transaction «Obtaining data from the unified database» (P.MM.08.TRN.004)
18. The common process transaction «Obtaining data from the unified database» (P.MM.08.TRN.004) is executed to provide corresponding data by the respondent upon the request of the initiator. The execution schema of the specified common process transaction is presented in Figure 7. The parameters of the common process transaction are given in Table 8.

Fig. 7. Execution schema of the common process transaction «Obtaining data from the unified database» (P.MM.08.TRN.004)
Table 8
Description of the common process transaction «Obtaining data from the unified database» (P.MM.08.TRN.004)
| No. | Mandatory element | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.TRN.004 |
| 2 | Name of the common process transaction | obtaining data from the unified database |
| 3 | Template of the common process transaction | request/response |
| 4 | Initiating role | initiator |
| 5 | Initiating operation | request and receipt of data from the unified database |
| 6 | Responding role | respondent |
| 7 | Receiving operation | provision of data from the unified database |
| 8 | Result of executing the common process transaction | unified database (P.MM.08.BEN.001): data are provided unified database (P.MM.08.BEN.001): data are absent |
| 9 | Parameters of the common process transaction: - time to confirm receipt - time to confirm acceptance for processing - response waiting time - authorization flag - number of retries | - 20 min 1 h yes 3 |
| 10 | Messages of the common process transaction: initiating message response message | request for data from the unified database (P.MM.08.MSG.006) data from the unified database (P.MM.08.MSG.007) notification on the absence of data in the unified database (P.MM.08.MSG.008) |
| 11 | Parameters of common process transaction messages: - EDS flag - transmission of an electronic document with an incorrect EDS | no - |
5. Common process transaction «Obtaining modified data from the unified database» (P.MM.08.TRN.005)
19. The common process transaction «Obtaining modified data from the unified database» (P.MM.08.TRN.005) is executed to provide corresponding data by the respondent upon the request of the initiator. The execution schema of the specified common process transaction is presented in Figure 8. The parameters of the common process transaction are given in Table 9.

Fig. 8. Execution schema of the common process transaction «Obtaining modified data from the unified database» (P.MM.08.TRN.005)
Table 9
Description of the common process transaction «Obtaining modified data from the unified database» (P.MM.08.TRN.005)
| No. | Mandatory element | Description |
|---|---|---|
| 1 | Code designation | P.MM.08.TRN.005 |
| 2 | Name of the common process transaction | obtaining modified data from the unified database |
| 3 | Template of the common process transaction | request/response |
| 4 | Initiating role | initiator |
| 5 | Initiating operation | request and receipt of modified data from the unified database |
| 6 | Responding role | respondent |
| 7 | Receiving operation | provision of modified data from the unified database |
| 8 | Result of executing the common process transaction | unified database (P.MM.08.BEN.001): modified data are provided unified database (P.MM.08.BEN.001): modified data are absent |
| 9 | Parameters of the common process transaction: - time to confirm receipt - time to confirm acceptance for processing - response waiting time - authorization flag - number of retries | - 20 min 1 h yes 3 |
| 10 | Messages of the common process transaction: initiating message response message | request for data from the unified database (P.MM.08.MSG.009) data from the unified database (P.MM.08.MSG.010) notification on the absence of data in the unified database (P.MM.08.MSG.011) |
| 11 | Parameters of common process transaction messages: - EDS flag - transmission of an electronic document with an incorrect EDS | no - |
VIII. Procedure for actions in non-standard situations
20. During information interaction within the framework of the common process, non-standard situations are likely when data processing cannot be carried out in a normal mode. Non-standard situations arise in the event of technical failures, timeouts, and in other cases. For a common process participant to receive comments on the causes of a non-standard situation and recommendations for its resolution, there is an option to send a corresponding request to the support service of the integrated information system of foreign and mutual trade. General recommendations for resolving a non-standard situation are given in Table 10.
21. The competent authority of the Member State checks the message regarding which an error notification was received for compliance with the Description of Formats and Structures of Electronic Documents and Data and the requirements for message control specified in Section IX of this Regulation. If non-compliance with the specified requirements is detected, the competent authority of the Member State takes all necessary measures to eliminate the detected error. If no non-compliances are detected, the competent authority of the Member State sends a message describing this non-standard situation to the support service of the integrated information system of foreign and mutual trade.
Table 10
Actions in non-standard situations
| Code of the non-standard situation | Description of the non-standard situation | Causes of the non-standard situation | Description of actions upon occurrence of a non-standard situation |
|---|---|---|---|
| P.EXC.002 | the initiator of the two-way common process transaction did not receive a response message after the expiration of the agreed number of retries | technical failures in the transport system or a systematic software error | it is necessary to send a request to the technical support service of the national segment in which the message was generated |
| P.EXC.004 | the initiator of the common process transaction received an error notification | reference books and classifiers are not synchronized or XML schemas of electronic documents (data) are not updated | the initiator of the common process transaction needs to synchronize the used reference books and classifiers or update the XML schemas of electronic documents (data). If the reference books and classifiers are synchronized, and the XML schemas of electronic documents (data) are updated, it is necessary to send a request to the support service of the receiving participant |
IX. Requirements for filling out electronic documents and data
22. The requirements for filling out the attributes of the electronic documents (data) «Data on the results of monitoring the safety, quality, and efficacy of medical devices» (R.HC.MM.08.001), transmitted in the message «Data on the results of monitoring the safety of medical devices for inclusion into the unified database» (P.MM.08.MSG.001), are given in Table 11.
Table 11
Requirements for filling out the attributes of the electronic documents (data) «Data on the results of monitoring the safety, quality, and efficacy of medical devices» (R.HC.MM.08.001), transmitted in the message «Data on the results of monitoring the safety of medical devices for inclusion into the unified database» (P.MM.08.MSG.001)
| Requirement code | Requirement formulation |
|---|---|
| 1 | 1 attribute «Data on monitoring of medical devices» (hccdo:MonitoringProductDetails) must be transmitted in the electronic message |
| 2 | the attribute «Start date and time» (csdo:StartDateTime) must be populated |
| 3 | the attribute «End date and time» (csdo:EndDateTime) is not populated |
| 4 | the unified database must not contain data on the results of monitoring the safety, quality, and efficacy of medical devices with the same values of the following attributes: «Country code» (csdo:UnifiedCountryCode); «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode), as well as «Document number» (csdo:DocId), «Document date» (csdo:DocCreationDate) within any of the complex attributes «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails), «Data on a corrective action for the safety of a medical device» (hccdo:CorrectionSafetyMedicalProductDetails), «Data on post-registration clinical monitoring of the safety and efficacy of a medical device» (hccdo:PostRegisrationClinicalMonitoringMedicalProductDetails) |
| 5 | the value of the attribute «Classifier identifier» (attribute codeListId) within the complex attribute «Country code» (csdo:UnifiedCountryCode) must contain the code designation of the classifier of countries of the world specified in Section VII of the Rules for Information Interaction |
| 6 | the value of the attribute «Country code» (csdo:UnifiedCountryCode) must correspond to the country code of the classifier of countries of the world containing the list of codes and names of countries of the world in accordance with the ISO 3166-1 standard |
| 7 | the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) or the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) within the complex attribute «Data on monitoring of medical devices» (hccdo:MonitoringProductDetails) must be populated |
| 8 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Business entity identifier» (csdo:BusinessEntityId) within it must be populated |
| 9 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Country code» (csdo:UnifiedCountryCode) within it must be populated |
| 10 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Name of the business entity» (csdo:BusinessEntityName) within it must be populated |
| 11 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Address» (ccdo:SubjectAddresDetails) within it must be populated |
| 12 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Country code» (csdo:UnifiedCountryCode) within the complex attribute «Address» (ccdo:SubjectAddresDetails) within it must be populated |
| 13 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the value of the attribute «Address type code» (csdo:AddressKindCode) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must correspond to the value «actual address» |
| 14 | in case the attribute «Address type code» (csdo:AddressKindCode) is populated, its value must correspond to one of the following values: 1 - «registration address»; 2 - «actual address»; 3 - «postal address» |
| 15 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «City» (csdo:CityName) or «Settlement» (csdo:SettlementName) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 16 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Street» (csdo:StreetName) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 17 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Building number» (csdo:BuildingNumberId) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 18 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Contact attribute» (ccdo:CommunicationDetails) within it must be populated |
| 19 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Code of the organizational and legal form» (csdo:BusinessEntityTypeCode) or the attribute «Name of the organizational and legal form» (csdo:BusinessEntityTypeName) must be populated |
| 20 | in case the attribute «Code of the organizational and legal form» (csdo:BusinessEntityTypeCode) is populated, the value of the attribute «Classifier identifier» (attribute codeListId) within it must contain the code designation of the classifier of organizational and legal forms specified in Section VII of the Rules for Information Interaction |
| 21 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Contact attribute» (ccdo:CommunicationDetails) must contain at least 1 value containing the value of the attribute «Communication type code» (csdo:CommunicationChannelCode) or «Name of the communication type» (csdo:CommunicationChannelName) corresponding to the communication type «telephone» |
| 22 | in case the value of the attribute «Communication type code» (csdo:CommunicationChannelCode) or «Name of the communication type» (csdo:CommunicationChannelName) corresponds to the value «e-mail», the value of the attribute «Communication channel identifier» (csdo:CommunicationChannelId) must match the pattern «.+@.+\..+» |
| 23 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Business entity identifier» (csdo:BusinessEntityId) within it must be populated |
| 24 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Code of the organizational and legal form» (csdo:BusinessEntityTypeCode) or the attribute «Name of the organizational and legal form» (csdo:BusinessEntityTypeName) must be populated |
| 25 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Country code» (csdo:UnifiedCountryCode) within it must be populated |
| 26 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Name of the business entity» (csdo:BusinessEntityName) within it must be populated |
| 27 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 28 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Country code» (csdo:UnifiedCountryCode) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 29 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the value of the attribute «Address type code» (csdo:AddressKindCode) within it must correspond to the value «actual address» |
| 30 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, one of the attributes «City» (csdo:CityName) or «Settlement» (csdo:SettlementName) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 31 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Street» (csdo:StreetName) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 32 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Building number» (csdo:BuildingNumberId) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 33 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Contact attribute» (ccdo:CommunicationDetails) within it must be populated |
| 34 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Contact attribute» (ccdo:CommunicationDetails) must contain at least 1 value containing the value of the attribute «Communication type code» (csdo:CommunicationChannelCode) or «Name of the communication type» (csdo:CommunicationChannelName) corresponding to the communication type «telephone» |
| 35 | in case the value of the attribute «Potential risk class of the medical device» (hcsdo:RiskClassCode) corresponds to the value «1 - low degree» or «2a - medium degree», the value of the attribute «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode) must not correspond to the value «03 - report on post-registration clinical monitoring of the safety and efficacy of a medical device» |
| 36 | in case the attribute «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode) corresponds to the value «01 - report on an adverse event (incident)», the attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) must be populated |
| 37 | in case the attribute «Address» (ccdo:SubjectAddressDetails) within the complex attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) is populated, the attribute «Address type code» (csdo:AddressKindCode) within it must be populated |
| 38 | in case the attribute «Address» (ccdo:SubjectAddressDetails) within the attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) is populated, the value of the attribute «Address type code» (csdo:AddressKindCode) within it must correspond to the value «actual address» |
| 39 | in case the attribute «Address» (ccdo:SubjectAddressDetails) within the complex attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) is populated, the attribute «Country code» (csdo:UnifiedCountryCode) within it must be populated |
| 40 | in case the attribute «Address» (ccdo:SubjectAddressDetails) within the complex attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) is populated, the attribute «City» (csdo:CityName) or «Settlement» (csdo:SettlementName) within it must be populated |
| 41 | in case the value of the attribute «Code of the user type at the time of occurrence of an adverse event (incident)» (hcsdo:UserKindTimeAdverseIncidentCode) corresponds to the value «other», the attribute «User at the time of occurrence of an adverse event (incident)» (hcsdo:UserKindTimeAdverseIncidentName) must be populated |
| 42 | if the attribute «Communication type code» (csdo:CommunicationChannelCode) is populated, its value must correspond to one of the following values: AO - «website address on the Internet»; TE - «telephone»; EM - «e-mail»; FX - «telefax» |
| 43 | in case the value of the attribute «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode) corresponds to the value «01 - report on an adverse event (incident)» and the value of the attribute «Code of the manufacturer's report type on the safety of a medical device» (hcsdo:ReportCorrectInfluenceTypeCode) within the complex attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) corresponds to the value «03 - combined initial and final report» or «04 - final report», the attribute «Data on the results of the investigation of an adverse event (incident) (hccdo:ResultsInvestigationMedicalProductDetails)» must be populated |
| 44 | in case the attribute «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode) corresponds to the value «02 - report on a corrective action for the safety of a medical device», the attribute «Data on a corrective action for the safety of a medical device» (hccdo:CorrectionSafetyMedicalProductDetails) must be populated |
| 45 | in case the attribute «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode) corresponds to the value «03 - report on post-registration clinical monitoring of the safety and efficacy of a medical device», the attribute «Data on post-registration clinical monitoring of the safety and efficacy of a medical device» (hccdo:PostRegisrationClinicalMonitoringMedicalProductDetails) must be populated |
| 46 | the attribute «Scheme of post-registration clinical monitoring» (hcsdo:SchemePostRegistrationClinicalMonitoringText) or the attribute «Document in PDF format» (hcsdo:PdfBinaryText) within the complex attribute «Data on the scheme of post-registration clinical monitoring» (hccdo:SchemePostRegistrationClinicalMonitoringDetails) must be populated |
| 47 | the attribute «Clinical data obtained during the reporting period» (hcsdo:ClinicalText) or the attribute «Document in PDF format» (hcsdo:PdfBinaryText) within the complex attribute «Data on clinical data obtained during the reporting period» (hccdo:ClinicalDataDetails) must be populated |
23. The requirements for filling out the attributes of the electronic documents (data) «Data on the results of monitoring the safety, quality, and efficacy of medical devices» (R.HC.MM.08.001), transmitted in the message «Data on the results of monitoring the safety of medical devices for modification in the unified database» (P.MM.08.MSG.002), are given in Table 12.
Table 12
Requirements for filling out the attributes of the electronic documents (data) «Data on the results of monitoring the safety, quality, and efficacy of medical devices» (R.HC.MM.08.001), transmitted in the message «Data on the results of monitoring the safety of medical devices for modification in the unified database» (P.MM.08.MSG.002)
| Requirement code | Requirement formulation |
|---|---|
| 1 | 1 attribute «Data on monitoring of medical devices» (hccdo:MonitoringProductDetails) must be transmitted in the electronic message |
| 2 | the attribute «Start date and time» (csdo:StartDateTime) must be populated |
| 3 | the attribute «End date and time» (csdo:EndDateTime) is not populated |
| 4 | the unified database must contain data on the results of monitoring the safety, quality, and efficacy of medical devices with the same values of the following attributes: «Country code» (csdo:UnifiedCountryCode); «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode), as well as «Document number» (csdo:DocId), «Document date» (csdo:DocCreationDate) within any of the complex attributes «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails), «Data on a corrective action for the safety of a medical device» (hccdo:CorrectionSafetyMedicalProductDetails), «Data on post-registration clinical monitoring of the safety and efficacy of a medical device» (hccdo:PostRegisrationClinicalMonitoringMedicalProductDetails) |
| 5 | the value of the attribute «Classifier identifier» (attribute codeListId) within the complex attribute «Country code» (csdo:UnifiedCountryCode) must contain the code designation of the classifier of countries of the world specified in Section VII of the Rules for Information Interaction |
| 6 | the value of the attribute «Country code» (csdo:UnifiedCountryCode) must correspond to the country code of the classifier of countries of the world in accordance with ISO 3166-1 |
| 7 | within the complex attribute «Data on monitoring of medical devices» (hccdo:MonitoringProductDetails), the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) or «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) must be populated |
| 8 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Business entity identifier» (csdo:BusinessEntityId) within it must be populated |
| 9 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Country code» (csdo:UnifiedCountryCode) within it must be populated |
| 10 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Name of the business entity» (csdo:BusinessEntityName) within it must be populated |
| 11 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Address» (ccdo:SubjectAddresDetails) within it must be populated |
| 12 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Country code» (csdo:UnifiedCountryCode) within the complex attribute «Address» (ccdo:SubjectAddresDetails) within it must be populated |
| 13 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the value of the attribute «Address type code» (csdo:AddressKindCode) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must correspond to the value «actual address» |
| 14 | in case the attribute «Address type code» (csdo:AddressKindCode) within any complex attributes is populated, its value must correspond to one of the following values: 1 - «registration address»; 2 - «actual address»; 3 - «postal address» |
| 15 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «City» (csdo:CityName) or «Settlement» (csdo:SettlementName) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 16 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Street» (csdo:StreetName) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 17 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Building number» (csdo:BuildingNumberId) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 18 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Contact attribute» (ccdo:CommunicationDetails) within it must be populated |
| 19 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Contact attribute» (ccdo:CommunicationDetails) must contain at least 1 value containing the value of the attribute «Communication type code» (csdo:CommunicationChannelCode) or «Name of the communication type» (csdo:CommunicationChannelName) corresponding to the communication type «telephone» |
| 20 | in case the attribute «Data on the manufacturer» (hccdo:ManufacturingAuthorizationHolderDetailsV2) is populated, the attribute «Code of the organizational and legal form» (csdo:BusinessEntityTypeCode) or the attribute «Name of the organizational and legal form» (csdo:BusinessEntityTypeName) must be populated |
| 21 | in case the attribute «Code of the organizational and legal form» (csdo:BusinessEntityTypeCode) is populated, the value of the attribute «Classifier identifier» (attribute codeListId) within it must contain the code designation of the classifier of organizational and legal forms specified in Section VII of the Rules for Information Interaction |
| 22 | in case the value of the attribute «Communication type code» (csdo:CommunicationChannelCode) or «Name of the communication type» (csdo:CommunicationChannelName) corresponds to the value «e-mail», the value of the attribute «Communication channel identifier» (csdo:CommunicationChannelId) must match the pattern «.+@.+\..+» |
| 23 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Business entity identifier» (csdo:BusinessEntityId) within it must be populated |
| 24 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Country code» (csdo:UnifiedCountryCode) within it must be populated |
| 25 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Name of the business entity» (csdo:BusinessEntityName) within it must be populated |
| 26 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 27 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Country code» (csdo:UnifiedCountryCode) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 28 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the value of the attribute «Address type code» (csdo:AddressKindCode) within it must correspond to the value «actual address» |
| 29 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, one of the attributes «City» (csdo:CityName) or «Settlement» (csdo:SettlementName) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 30 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Street» (csdo:StreetName) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 31 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Building number» (csdo:BuildingNumberId) within the complex attribute «Address» (ccdo:SubjectAddressDetails) within it must be populated |
| 32 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Contact attribute» (ccdo:CommunicationDetails) within it must be populated |
| 33 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Contact attribute» (ccdo:CommunicationDetails) must contain at least 1 value containing the value of the attribute «Communication type code» (csdo:CommunicationChannelCode) or «Name of the communication type» (csdo:CommunicationChannelName) corresponding to the communication type «telephone» |
| 34 | in case the attribute «Data on the authorized representative of the manufacturer of the medical device» (hccdo:MedicalProductManufacturerAgentDetails) is populated, the attribute «Code of the organizational and legal form» (csdo:BusinessEntityTypeCode) or the attribute «Name of the organizational and legal form» (csdo:BusinessEntityTypeName) must be populated |
| 35 | in case the value of the attribute «Potential risk class of the medical device» (hcsdo:RiskClassCode) corresponds to the value «1 - low degree» or «2a - medium degree», the value of the attribute «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode) must not correspond to the value «03 - report on post-registration clinical monitoring of the safety and efficacy of a medical device» |
| 36 | in case the attribute «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode) corresponds to the value «01 - report on an adverse event (incident)», the attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) must be populated |
| 37 | in case the attribute «Address» (ccdo:SubjectAddressDetails) within the complex attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) is populated, the attribute «Address type code» (csdo:AddressKindCode) within it must be populated |
| 38 | in case the attribute «Address» (ccdo:SubjectAddressDetails) within the attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) is populated, the value of the attribute «Address type code» (csdo:AddressKindCode) within it must correspond to the value «actual address» |
| 39 | in case the attribute «Address» (ccdo:SubjectAddressDetails) within the complex attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) is populated, the attribute «Country code» (csdo:UnifiedCountryCode) within it must be populated |
| 40 | in case the attribute «Address» (ccdo:SubjectAddressDetails) within the complex attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) is populated, the attribute «City» (csdo:CityName) or «Settlement» (csdo:SettlementName) must be populated |
| 41 | in case the value of the attribute «Code of the user type at the time of occurrence of an adverse event (incident)» (hcsdo:UserKindTimeAdverseIncidentCode) corresponds to the value «other», the attribute «User at the time of occurrence of an adverse event (incident)» (hcsdo:UserKindTimeAdverseIncidentName) must be populated |
| 42 | if the attribute «Communication type code» (csdo:CommunicationChannelCode) is populated, its value must correspond to one of the following values: AO - «website address on the Internet»; TE - «telephone»; EM - «e-mail»; FX - «telefax» |
| 43 | in case the value of the attribute «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode) corresponds to the value «01 - report on an adverse event (incident)» and the value of the attribute «Code of the manufacturer's report type on the safety of a medical device» (hcsdo:ReportCorrectInfluenceTypeCode) within the complex attribute «Data on an adverse event (incident)» (hccdo:AdverseIncidentMedicalProductDetails) corresponds to the value «03 - combined initial and final report» or «04 - final report», the attribute «Data on the results of the investigation of an adverse event (incident) (hccdo:ResultsInvestigationMedicalProductDetails)» must be populated |
| 44 | in case the attribute «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode) corresponds to the value «02 - report on a corrective action for the safety of a medical device», the attribute «Data on a corrective action for the safety of a medical device» (hccdo:CorrectionSafetyMedicalProductDetails) must be populated |
| 45 | in case the attribute «Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device» (hcsdo:MedicalProductMonitoringDocKindCode) corresponds to the value «03 - report on post-registration clinical monitoring of the safety and efficacy of a medical device», the attribute «Data on post-registration clinical monitoring of the safety and efficacy of a medical device» (hccdo:PostRegisrationClinicalMonitoringMedicalProductDetails) must be populated |
| 46 | the attribute «Scheme of post-registration clinical monitoring» (hcsdo:SchemePostRegistrationClinicalMonitoringText) or the attribute «Document in PDF format» (hcsdo:PdfBinaryText) within the complex attribute «Data on the scheme of post-registration clinical monitoring» (hccdo:SchemePostRegistrationClinicalMonitoringDetails) must be populated |
| 47 | the attribute «Clinical data obtained during the reporting period» (hcsdo:ClinicalText) or the attribute «Document in PDF format» (hcsdo:PdfBinaryText) within the complex attribute «Data on clinical data obtained during the reporting period» (hccdo:ClinicalDataDetails) must be populated |
Approved
by Decision of the Board
of the Eurasian Economic Commission
of August 30, 2016 No. 94
DESCRIPTION
OF FORMATS AND STRUCTURES OF ELECTRONIC DOCUMENTS AND DATA USED FOR THE IMPLEMENTATION, BY MEANS OF THE INTEGRATED INFORMATION SYSTEM OF FOREIGN AND MUTUAL TRADE, OF THE COMMON PROCESS «FORMATION, MAINTENANCE AND USE OF THE UNIFIED INFORMATION DATABASE FOR MONITORING THE SAFETY, QUALITY AND EFFICACY OF MEDICAL DEVICES»
I. General Provisions
1. This Description is developed in accordance with the following acts comprising the law of the Eurasian Economic Union (hereinafter - the Union):
Treaty on the Eurasian Economic Union of May 29, 2014;
Decision of the Board of the Eurasian Economic Commission of November 6, 2014 No. 200 «On Technological Documents Regulating Information Interaction in the Implementation of Common Processes by Means of the Integrated Information System of Foreign and Mutual Trade»;
Decision of the Board of the Eurasian Economic Commission of January 27, 2015 No. 5 «On Approval of the Rules for Electronic Data Exchange in the Integrated Information System of Foreign and Mutual Trade»;
Decision of the Board of the Eurasian Economic Commission of April 14, 2015 No. 29 «On the List of Common Processes within the Eurasian Economic Union and the Introduction of an Amendment to Decision of the Board of the Eurasian Economic Commission of August 19, 2014 No. 132»;
Decision of the Board of the Eurasian Economic Commission of June 9, 2015 No. 63 «On the Methodology for the Analysis, Optimization, Harmonization, and Description of Common Processes within the Eurasian Economic Union»;
Decision of the Board of the Eurasian Economic Commission of September 28, 2015 No. 125 «On Approval of the Regulation on the Exchange of Electronic Documents during Transboundary Interaction of Public Authorities of the Member States of the Eurasian Economic Union Among Themselves and with the Eurasian Economic Commission».
II. Scope of Application
2. This Description determines the requirements for the formats and structures of electronic documents and data used during information interaction within the framework of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices» (hereinafter - the common process).
3. This Description is applied during the design, development, and refinement of information system components in the implementation of the common process procedures by means of the integrated information system of foreign and mutual trade (hereinafter - the integrated system).
4. The description of the formats and structures of electronic documents and data is provided in tabular form indicating the complete attribute composition, taking into account hierarchy levels down to simple (atomic) attributes.
5. The table describes a one-to-one correspondence of the attributes of electronic documents (data) (hereinafter - attributes) and the data model elements.
6. The following fields (columns) are formed in the table:
«hierarchical number» - ordinal number of the attribute;
«attribute name» - established or official verbal designation of the attribute;
«attribute description» - text explaining the meaning (semantics) of the attribute;
«identifier» - identifier of the data element in the data model corresponding to the attribute;
«value domain» - verbal description of the possible values of the attribute;
«Mult.» - multiplicity of attributes: obligatoriness (optionality) and the number of possible repetitions of the attribute.
7. The following notations are used to indicate the multiplicity of attributes:
1 - the attribute is mandatory, repetitions are not allowed;
n - the attribute is mandatory, must be repeated n times (n > 1);
1..* - the attribute is mandatory, can be repeated without restrictions;
n..* - the attribute is mandatory, must be repeated at least n times (n > 1);
n..m - the attribute is mandatory, must be repeated at least n times and no more than m times (n > 1, m > n);
0..1 - the attribute is optional, repetitions are not allowed;
0..* - the attribute is optional, can be repeated without restrictions;
0..m - the attribute is optional, can be repeated no more than m times (m > 1).
III. Basic Concepts
8. For the purposes of this Description, concepts are used that mean the following:
«Member State» - a state that is a member of the Union;
«attribute» - a data element of an electronic document (data) that is considered indivisible in a specific context.
The concepts of «basic data model», «data model», «domain data model», «domain», and «register of structures of electronic documents and data» used in this Description are applied in the meanings defined by the Methodology for the Analysis, Optimization, Harmonization, and Description of Common Processes within the Eurasian Economic Union, approved by Decision of the Board of the Eurasian Economic Commission of June 9, 2015 No. 63.
Other concepts used in this Description are applied in the meanings defined in paragraph 4 of the Rules for information interaction in the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices», approved by Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94.
In Tables 4, 7, and 10 of this Description, the Regulation on Information Interaction refers to the Regulation on information interaction between the competent authorities of the Member States of the Eurasian Economic Union and the Eurasian Economic Commission in the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices», approved by Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94.
IV. Structures of Electronic Documents and Data
9. The list of structures of electronic documents and data is given in Table 1.
Table 1
List of structures of electronic documents and data
| No. | Identifier | Name | Namespace |
|---|---|---|---|
| 1. Structures of electronic documents and data in the basic model | |||
| 1.1 | R.006 | notification on the processing result | urn:EEC:R:ProcessingResultDetails:vY.Y.Y |
| 1.2 | R.007 | state of updating of a shared resource | urn:EEC:R:ResourceStatusDetails:vY.Y.Y |
| 2. Structures of electronic documents and data in the "Healthcare" domain | |||
| 2.1 | R.HC.MM.08.001 | data on the results of monitoring the safety, quality, and efficacy of medical devices | urn:EEC:R:HC:MM:08:MedicineProductDocDetails:v1.0.0 |
The symbols «Y.Y.Y» in the namespaces of the electronic document (data) structure correspond to the version number of the electronic document (data) structure, determined in accordance with the version number of the basic data model used in the development of the technical schema of the electronic document (data) structure in accordance with paragraph 2 of Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94.
1. Structures of electronic documents and data in the basic model
10. The description of the structure of the electronic document (data) «Notification on the processing result» (R.006) is given in Table 2.
Table 2
Description of the structure of the electronic document (data) «Notification on the processing result» (R.006)
| No. | Element designation | Description |
|---|---|---|
| 1 | Name | notification on the processing result |
| 2 | Identifier | R.006 |
| 3 | Version | Y.Y.Y |
| 4 | Definition | data on the result of processing the request by the respondent |
| 5 | Usage | - |
| 6 | Namespace identifier | urn:EEC:R:ProcessingResultDetails:vY.Y.Y |
| 7 | Root element of the XML document | ProcessingResultDetails |
| 8 | XML schema file name | EEC_R_ProcessingResultDetails_vY.Y.Y.xsd |
The symbols «Y.Y.Y» in the namespaces of the electronic document (data) structure correspond to the version number of the electronic document (data) structure, determined in accordance with the version number of the basic data model used in the development of the technical schema of the electronic document (data) structure in accordance with paragraph 2 of Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94.
11. The imported namespaces are given in Table 3.
Table 3
Imported namespaces
| No. | Namespace identifier | Prefix |
|---|---|---|
| 1 | urn:EEC:M:ComplexDataObjects:vX.X.X | ccdo |
| 2 | urn:EEC:M:SimpleDataObjects:vX.X.X | csdo |
The symbols «X.X.X» in the imported namespaces correspond to the version number of the basic data model used in the development of the technical schema of the electronic document (data) structure in accordance with paragraph 2 of Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94.
12. The attribute composition of the structure of the electronic document (data) «Notification on the processing result» (R.006) is given in Table 4.
Table 4
Attribute composition of the structure of the electronic document (data) «Notification on the processing result» (R.006)
| Attribute name | Attribute description | Identifier | Data type | Mult. |
|---|---|---|---|---|
| 1. Header of the electronic document (data) (ccdo:EDocHeader) | set of technological attributes of the electronic document (data) | M.CDE.90001 | ccdo:EDocHeaderType (M.CDT.90001) Determined by the value domains of nested elements | 1 |
| 1.1. Code of the common process message (csdo:InfEnvelopeCode) | code designation of the common process message | M.SDE.90010 | csdo:InfEnvelopeCodeType (M.SDT.90004) Code value in accordance with the Regulation on Information Interaction. Pattern: P\.[A-Z]{2}\.[0-9]{2}\.MSG\.[0-9]{3} | 1 |
| 1.2. Code of the electronic document (data) (csdo:EDocCode) | code designation of the electronic document (data) in accordance with the register of structures of electronic documents and data | M.SDE.90001 | csdo:EDocCodeType (M.SDT.90001) Code value in accordance with the register of structures of electronic documents and data. Pattern: R(\.[A-Z]{2}\.[A-Z]{2}\.[0-9]{2})?\.[0-9]{3} | 1 |
| 1.3. Identifier of the electronic document (data) (csdo:EDocId) | string of characters uniquely identifying the electronic document (data) | M.SDE.90007 | csdo:UniversallyUniqueIdType (M.SDT.90003) Identifier value in accordance with ISO/IEC 9834-8. Pattern: [0-9a-fA-F]{8}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{12} | 1 |
| 1.4. Identifier of the source electronic document (data) (csdo:EDocRefId) | identifier of the electronic document (data) in response to which this electronic document (data) was generated | M.SDE.90008 | csdo:UniversallyUniqueIdType (M.SDT.90003) Identifier value in accordance with ISO/IEC 9834-8. Pattern: [0-9a-fA-F]{8}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{12} | 0..1 |
| 1.5. Date and time of the electronic document (data) (csdo:EDocDateTime) | date and time of creation of the electronic document (data) | M.SDE.90002 | bdt:DateTimeType (M.BDT.00006) Designation of date and time in accordance with GOST ISO 8601-2001 | 1 |
| 1.6. Language code (csdo:LanguageCode) | code designation of the language | M.SDE.00051 | csdo:LanguageCodeType (M.SDT.00051) Two-letter language code in accordance with ISO 639-1. Pattern: [a-z]{2} | 0..1 |
| 2. Date and time (csdo:EventDateTime) | date and time of completion of data processing | M.SDE.00132 | bdt:DateTimeType (M.BDT.00006) Designation of date and time in accordance with GOST ISO 8601-2001 | 1 |
| 3. Code of the processing result (csdo:ProcessingResultV2Code) | code designation of the result of processing the received electronic document (data) by the information system of the common process participant | M.SDE.90014 | csdo:ProcessingResultCodeV2Type (M.SDT.90006) Code value in accordance with the classifier of processing results for electronic documents and data | 1 |
| 4. Description (csdo:DescriptionText) | description of the data processing result in free form | M.SDE.00002 | csdo:Text4000Type (M.SDT.00088) String of characters. Min. length: 1. Max. length: 4000 | 0..1 |
13. The description of the structure of the electronic document (data) «State of updating of a shared resource» (R.007) is given in Table 5.
Table 5
Description of the structure of the electronic document (data) «State of updating of a shared resource» (R.007)
| No. | Element designation | Description |
|---|---|---|
| 1 | Name | state of updating of a shared resource |
| 2 | Identifier | R.007 |
| 3 | Version | Y.Y.Y |
| 4 | Definition | data for updating a shared resource |
| 5 | Usage | used to request the date and time of updating of a shared resource and to respond to this request, as well as to request actual or complete (modified, updated) data from a shared resource |
| 6 | Namespace identifier | urn:EEC:R:ResourceStatusDetails:vY.Y.Y |
| 7 | Root element of the XML document | ResourceStatusDetails |
| 8 | XML schema file name | EEC_R_ResourceStatusDetails_vY.Y.Y.xsd |
The symbols «Y.Y.Y» in the namespaces of the electronic document (data) structure correspond to the version number of the electronic document (data) structure, determined in accordance with the version number of the basic data model used in the development of the technical schema of the electronic document (data) structure in accordance with paragraph 2 of Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94.
14. The imported namespaces are given in Table 6.
Table 6
Imported namespaces
| No. | Namespace identifier | Prefix |
|---|---|---|
| 1 | urn:EEC:M:ComplexDataObjects:vX.X.X | ccdo |
| 2 | urn:EEC:M:SimpleDataObjects:vX.X.X | csdo |
The symbols «X.X.X» in the imported namespaces correspond to the version number of the basic data model used in the development of the technical schema of the electronic document (data) structure in accordance with paragraph 2 of Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94.
15. The attribute composition of the structure of the electronic document (data) «State of updating of a shared resource» (R.007) is given in Table 7.
Table 7
Attribute composition of the structure of the electronic document (data) «State of updating of a shared resource» (R.007)
| Attribute name | Attribute description | Identifier | Data type | Mult. |
|---|---|---|---|---|
| 1. Header of the electronic document (data) (ccdo:EDocHeader) | set of technological attributes of the electronic document (data) | M.CDE.90001 | ccdo:EDocHeaderType (M.CDT.90001) Determined by the value domains of nested elements | 1 |
| 1.1. Code of the common process message (csdo:InfEnvelopeCode) | code designation of the common process message | M.SDE.90010 | csdo:InfEnvelopeCodeType (M.SDT.90004) Code value in accordance with the Regulation on Information Interaction. Pattern: P\.[A-Z]{2}\.[0-9]{2}\.MSG\.[0-9]{3} | 1 |
| 1.2. Code of the electronic document (data) (csdo:EDocCode) | code designation of the electronic document (data) in accordance with the register of structures of electronic documents and data | M.SDE.90001 | csdo:EDocCodeType (M.SDT.90001) Code value in accordance with the register of structures of electronic documents and data. Pattern: R(\.[A-Z]{2}\.[A-Z]{2}\.[0-9]{2})?\.[0-9]{3} | 1 |
| 1.3. Identifier of the electronic document (data) (csdo:EDocId) | string of characters uniquely identifying the electronic document (data) | M.SDE.90007 | csdo:UniversallyUniqueIdType (M.SDT.90003) Identifier value in accordance with ISO/IEC 9834-8. Pattern: [0-9a-fA-F]{8}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{12} | 1 |
| 1.4. Identifier of the source electronic document (data) (csdo:EDocRefId) | identifier of the electronic document (data) in response to which this electronic document (data) was generated | M.SDE.90008 | csdo:UniversallyUniqueIdType (M.SDT.90003) Identifier value in accordance with ISO/IEC 9834-8. Pattern: [0-9a-fA-F]{8}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{12} | 0..1 |
| 1.5. Date and time of the electronic document (data) (csdo:EDocDateTime) | date and time of creation of the electronic document (data) | M.SDE.90002 | bdt:DateTimeType (M.BDT.00006) Designation of date and time in accordance with GOST ISO 8601-2001 | 1 |
| 1.6. Language code (csdo:LanguageCode) | code designation of the language | M.SDE.00051 | csdo:LanguageCodeType (M.SDT.00051) Two-letter language code in accordance with ISO 639-1. Pattern: [a-z]{2} | 0..1 |
| 2. Date and time of update (csdo:UpdateDateTime) | date and time of updating of a shared resource (register, list, database) | M.SDE.00079 | bdt:DateTimeType (M.BDT.00006) Designation of date and time in accordance with GOST ISO 8601-2001 | 0..1 |
| 3. Country code (csdo:UnifiedCountryCode) | code designation of the country that submitted data to the shared resource (register, list, database) | M.SDE.00162 | csdo:UnifiedCountryCodeType (M.SDT.00112) Value of the two-letter code in accordance with the classifier of countries of the world, which is determined by the attribute «Identifier of the reference book (classifier)». Pattern: [A-Z]{2} | 0..* |
| a) identifier of the reference book (classifier) (attribute codeListId) | designation of the reference book (classifier) in accordance with which the code is specified | - | csdo:ReferenceDataIdType (M.SDT.00091) Normalized string of characters containing no line break (#xA) or tab (#x9) characters. Min. length: 1. Max. length: 20 | 1 |
2. Structures of electronic documents and data in the «Healthcare» domain
16. The description of the structure of the electronic document (data) «Data on the results of monitoring the safety, quality, and efficacy of medical devices» (R.HC.MM.08.001) is given in Table 8.
Table 8
Description of the structure of the electronic document (data) «Data on the results of monitoring the safety, quality, and efficacy of medical devices» (R.HC.MM.08.001)
| No. | Element designation | Description |
|---|---|---|
| 1 | Name | data on the results of monitoring the safety, quality, and efficacy of medical devices |
| 2 | Identifier | R.HC.MM.08.001 |
| 3 | Version | 1.0.0 |
| 4 | Definition | data on the results of monitoring the safety, quality, and efficacy of medical devices |
| 5 | Usage | - |
| 6 | Namespace identifier | urn:EEC:R:HC:MM:08:MedicineProductDocDetails:v1.0.0 |
| 7 | Root element of the XML document | MedicineProductDocDetails |
| 8 | XML schema file name | EEC_R_HC_MM_08_MedicineProductDocDetails_v1.0.0.xsd |
17. The imported namespaces are given in Table 9.
Table 9
Imported namespaces
| No. | Namespace identifier | Prefix |
|---|---|---|
| 1 | urn:EEC:M:ComplexDataObjects:vX.X.X | ccdo |
| 2 | urn:EEC:M:HC:ComplexDataObjects:vX.X.X | hccdo |
| 3 | urn:EEC:M:HC:SimpleDataObjects:vX.X.X | hcsdo |
| 4 | urn:EEC:M:SimpleDataObjects:vX.X.X | csdo |
The symbols «X.X.X» in the imported namespaces correspond to the version number of the basic data model and the domain data model used in the development of the technical schema of the electronic document (data) structure in accordance with paragraph 2 of Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94.
18. The attribute composition of the structure of the electronic document (data) «Data on the results of monitoring the safety, quality, and efficacy of medical devices» (R.HC.MM.08.001) is given in Table 10.
Table 10
Attribute composition of the structure of the electronic document (data) «Data on the results of monitoring the safety, quality, and efficacy of medical devices» (R.HC.MM.08.001)
Table 10 contains a detailed attribute composition and is placed on the site in its original form (HTML-table with a full set of attributes). Due to its exceptional volume (over 100 rows with nested attributes), it is extracted into a separate file — part5b_table10.html
Approved
by Decision of the Board
of the Eurasian Economic Commission
of August 30, 2016 No. 94
PROCEDURE
FOR JOINING THE COMMON PROCESS «FORMATION, MAINTENANCE AND USE OF THE UNIFIED INFORMATION DATABASE FOR MONITORING THE SAFETY, QUALITY AND EFFICACY OF MEDICAL DEVICES»
I. General Provisions
1. This Procedure is developed in accordance with the following acts comprising the law of the Eurasian Economic Union (hereinafter - the Union):
Treaty on the Eurasian Economic Union of May 29, 2014;
Agreement on Common Principles and Rules for Circulation of Medical Devices (Medical Equipment and Medical Materials) within the Eurasian Economic Union of December 23, 2014;
Decision of the Supreme Eurasian Economic Council of December 23, 2014 No. 109 «On the Implementation of the Agreement on Common Principles and Rules for Circulation of Medical Devices (Medical Equipment and Medical Materials) within the Eurasian Economic Union»
Decision of the Board of the Eurasian Economic Commission of November 6, 2014 No. 200 «On Technological Documents Regulating Information Interaction in the Implementation of Common Processes by Means of the Integrated Information System of Foreign and Mutual Trade»;
Decision of the Board of the Eurasian Economic Commission of January 27, 2015 No. 5 «On Approval of the Rules for Electronic Data Exchange in the Integrated Information System of Foreign and Mutual Trade»;
Decision of the Board of the Eurasian Economic Commission of April 14, 2015 No. 29 «On the List of Common Processes within the Eurasian Economic Union and the Introduction of an Amendment to Decision of the Board of the Eurasian Economic Commission of August 19, 2014 No. 132»;
Decision of the Board of the Eurasian Economic Commission of June 9, 2015 No. 63 «On the Methodology for the Analysis, Optimization, Harmonization, and Description of Common Processes within the Eurasian Economic Union».
II. Scope of Application
2. This Procedure determines the requirements for the composition and content of the procedures for putting into operation the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices» (P.MM.08) (hereinafter - the common process) and for the joining of a new participant to the common process, as well as the requirements for the information interaction carried out during their execution.
III. Basic Concepts
3. For the purposes of this Procedure, concepts are used that mean the following:
«documents applied to ensure the functioning of the integrated system» - technical, technological, methodological, and organizational documents provided for by paragraph 30 of the Protocol on Information and Communication Technologies and Information Interaction within the Eurasian Economic Union (Annex No. 3 to the Treaty on the Eurasian Economic Union of May 29, 2014);
«technological documents» - documents included in the standard list of technological documents regulating information interaction in the implementation of the common process, as provided for in paragraph 1 of Decision of the Board of the Eurasian Economic Commission of November 6, 2014 No. 200.
Other concepts used in this Procedure are applied in the meanings defined by paragraph 4 of the Rules for information interaction in the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices», approved by Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94 (hereinafter - the Rules for Information Interaction).
IV. Interaction Participants
4. The roles of the interaction participants when they execute the procedures provided for by this Procedure are given in the table.
Table
Roles of the interaction participants
| No. | Name of the role | Description of the role | Participant performing the role |
|---|---|---|---|
| 1 | Joining participant of the common process | executes the procedures provided for by this Procedure | competent authority of a Member State of the Union |
| 2 | Administrator | coordinates the execution of procedures provided for by this Procedure and participates in testing the information interaction with the joining participant of the common process | The Eurasian Economic Commission |
V. Putting the Common Process into Operation
5. From the date of entry into force of Decision of the Board of the Eurasian Economic Commission of August 30, 2016 No. 94 «On technological documents regulating information interaction in the implementation, by means of the integrated information system of foreign and mutual trade, of the common process «Formation, maintenance and use of the unified information database for monitoring the safety, quality and efficacy of medical devices», the Member States of the Union (hereinafter - the Member States) under the coordination of the Eurasian Economic Commission (hereinafter - The Commission) shall commence the execution of the procedure for putting the common process into operation.
6. To put the common process into operation, the necessary activities determined by the procedure for joining the common process must be performed by the Member States in accordance with Section VI of this Procedure.
7. Based on the recommendations of the commission for conducting interstate tests of the integrated information system of foreign and mutual trade, the Board of The Commission adopts an order on putting the common process into operation.
8. The basis for adopting the recommendation of the commission for conducting interstate tests of the integrated information system of foreign and mutual trade regarding the readiness of the common process for operation may be the results of testing the information interaction between the information systems of one of the Member States and The Commission.
VI. Description of the Joining Procedure
9. After putting the common process into operation, new participants may join it by executing the procedure for joining the common process.
10. To join the common process, the joining participant of the common process must fulfill the requirements of the documents applied to ensure the functioning of the integrated system, technological documents, as well as the requirements of the legislation of the Member State regulating information interaction within the national segment of the Member State.
11. The execution of the procedure for joining a new participant to the common process includes:
a) informing The Commission by the Member State about the joining of a new participant to the common process (specifying the competent authority responsible for ensuring information interaction within the common process);
b) introducing into the regulatory legal acts of the Member State the changes necessary to fulfill the requirements of the technological documents (within 2 months from the start date of executing the joining procedure);
c) developing (refining), if necessary, the information system of the joining participant of the common process, including with regard to the use of electronic digital signature (electronic signature) tools compatible with the services of the trusted third party of the national segment of the Member State (within 3 months from the start date of executing the joining procedure);
d) connecting the information system of the joining participant of the common process to the national segment of the Member State, if such connection was not made earlier (within 3 months from the start date of executing the joining procedure);
e) receiving by the joining participant of the common process the reference books and classifiers distributed by the administrator as specified in the Rules for Information Interaction;
f) testing the information interaction between the information systems of the joining participants of the common process and the administrator for compliance with the requirements of the technological documents (within 6 months from the start date of executing the joining procedure).
Table 10
Attribute composition of the structure of the electronic document (data) «Data on the results of monitoring the safety, quality, and efficacy of medical devices» (R.HC.MM.08.001)
| Attribute name | Attribute description | Identifier | Data type | Mult. |
|---|---|---|---|---|
| 1. Header of the electronic document (data) (ccdo:EDocHeader) | set of technological attributes of the electronic document (data) | M.CDE.90001 | ccdo:EDocHeaderType (M.CDT.90001) Determined by the value domains of nested elements | 1 |
| 1.1. Code of the common process message (csdo:InfEnvelopeCode) | code designation of the common process message | M.SDE.90010 | csdo:InfEnvelopeCodeType (M.SDT.90004) Code value in accordance with the Regulation on Information Interaction. Pattern: P\.[A-Z]{2}\.[0-9]{2}\.MSG\.[0-9]{3} | 1 |
| 1.2. Code of the electronic document (data) (csdo:EDocCode) | code designation of the electronic document (data) in accordance with the register of structures of electronic documents and data | M.SDE.90001 | csdo:EDocCodeType (M.SDT.90001) Pattern: R(\.[A-Z]{2}\.[A-Z]{2}\.[0-9]{2})?\.[0-9]{3} | 1 |
| 1.3. Identifier of the electronic document (data) (csdo:EDocId) | string of characters uniquely identifying the electronic document (data) | M.SDE.90007 | csdo:UniversallyUniqueIdType (M.SDT.90003) Identifier value in accordance with ISO/IEC 9834-8. Pattern: [0-9a-fA-F]{8}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{12} | 1 |
| 1.4. Identifier of the source electronic document (data) (csdo:EDocRefId) | identifier of the electronic document (data) in response to which this electronic document (data) was generated | M.SDE.90008 | csdo:UniversallyUniqueIdType (M.SDT.90003) Pattern: [0-9a-fA-F]{8}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{4}-[0-9a-fA-F]{12} | 0..1 |
| 1.5. Date and time of the electronic document (data) (csdo:EDocDateTime) | date and time of creation of the electronic document (data) | M.SDE.90002 | bdt:DateTimeType (M.BDT.00006) Designation of date and time in accordance with GOST ISO 8601-2001 | 1 |
| 1.6. Language code (csdo:LanguageCode) | code designation of the language | M.SDE.00051 | csdo:LanguageCodeType (M.SDT.00051) Two-letter language code in accordance with ISO 639-1. Pattern: [a-z]{2} | 0..1 |
| 2. Data on monitoring of medical devices (hccdo:MonitoringProductDetails) | data on monitoring the safety, quality, and efficacy of medical devices | M.HC.CDE.00059 | hccdo:MonitoringProductDetailsType (M.HC.CDT.00050) Determined by the value domains of nested elements | 1..* |
| 2.1. Country code (csdo:UnifiedCountryCode) | code designation of the country | M.SDE.00162 | csdo:UnifiedCountryCodeType (M.SDT.00112) Pattern: [A-Z]{2} | 1 |
| a) identifier of the reference book (classifier) (attribute codeListId) | designation of the reference book (classifier) in accordance with which the code is specified | - | csdo:ReferenceDataIdType (M.SDT.00091) Min. length: 1. Max. length: 20 | 1 |
| 2.2. Name of the medical device (csdo:MedicalProductName) | name of the medical device | M.HC.SDE.00763 | csdo:Name500Type (M.SDT.00134) Min. length: 1. Max. length: 500 | 1 |
| 2.3. Factory (serial) number of the medical device (hcsdo:MedicalProductSerialId) | factory (serial) number of the medical device | M.HC.SDE.00581 | csdo:Id50Type (M.SDT.00093) Min. length: 1. Max. length: 50 | 0..1 |
| 2.4. Batch number (hcsdo:PartyNumberId) | batch number of the medical device | M.HC.SDE.00582 | csdo:Id50Type (M.SDT.00093) Min. length: 1. Max. length: 50 | 0..1 |
| 2.5. Data on the registration certificate of the medical device (hccdo:MedicalProductRegistrationCertificateV2Details) | data on the registration certificate of the medical device | M.HC.CDE.00781 | hccdo:MedicalProductRegistrationCertificateV2DetailsType (M.HC.CDT.00550) Determined by the value domains of nested elements | 1 |
| 2.5.1. Number of the registration certificate (hcsdo:RegistrationCertificateId) | number of the registration certificate | M.HC.SDE.00045 | hcsdo:RegistrationCertificateIdType (M.HC.SDT.00040) Min. length: 1. Max. length: 50 | 1 |
| 2.5.2. Registration date of the medical device (hcsdo:MedicalProductRegistrationDate) | registration date of the medical device | M.HC.SDE.00767 | bdt:DateType (M.BDT.00005) Designation of date in accordance with GOST ISO 8601-2001 | 1 |
| 2.5.3. Expiration date of the document (csdo:DocValidityDate) | expiration date of the registration certificate of the medical device | M.SDE.00052 | bdt:DateType (M.BDT.00005) | 0..1 |
| 2.5.4. Name of the competent authority of the reference state (hcsdo:ReferenceAuthorityName) | name of the competent authority of the reference state | M.HC.SDE.00717 | csdo:Name500Type (M.SDT.00134) Min. length: 1. Max. length: 500 | 0..1 |
| 2.6. Code of the medical device type (hcsdo:MedicalProductClassificationCode) | code designation of the medical device in accordance with the reference book «Nomenclature of Medical Devices of the Eurasian Economic Union» | M.HC.SDE.00447 | hcsdo:MedicalProductClassificationCodeType (M.HC.SDT.00580) Pattern: \d{6} | 0..1 |
| 2.7. Name of the medical device type (hcsdo:MedicalProductClassificationName) | name of the medical device type in accordance with the reference book «Nomenclature of Medical Devices of the Eurasian Economic Union» | M.HC.SDE.00448 | csdo:Name500Type (M.SDT.00134) Min. length: 1. Max. length: 500 | 0..1 |
| 2.8. Code of the potential risk class of the medical device (hcsdo:RiskClassCode) | code designation of the potential risk class of the medical device | M.HC.SDE.00032 | hcsdo:RiskClassCodeType (M.HC.SDT.00008) Possible values: 1 - low degree; 2a - medium degree; 2b - elevated degree; 3 - high degree | 1 |
| 2.9. Data on the manufacturer (hccdo:ManufacturingAuthorizationHolderDetailsV2) | data on the manufacturer of the medical device | M.HC.CDE.00304 | ccdo:BusinessEntityDetailsType (M.CDT.00061) Determined by the value domains of nested elements | 0..1 |
| 2.9.1. Country code (csdo:UnifiedCountryCode) | code designation of the registration country of the business entity | M.SDE.00162 | csdo:UnifiedCountryCodeType (M.SDT.00112) Pattern: [A-Z]{2} | 0..1 |
| a) identifier of the reference book (classifier) (attribute codeListId) | designation of the reference book (classifier) in accordance with which the code is specified | - | csdo:ReferenceDataIdType (M.SDT.00091) Min. length: 1. Max. length: 20 | 1 |
| 2.9.2. Name of the business entity (csdo:BusinessEntityName) | full name of the business entity or surname, first name and patronymic of an individual conducting business activities | M.SDE.00187 | csdo:Name300Type (M.SDT.00056) Min. length: 1. Max. length: 300 | 0..1 |
| 2.9.3. Brief name of the business entity (csdo:BusinessEntityBriefName) | abbreviated name of the business entity | M.SDE.00188 | csdo:Name120Type (M.SDT.00055) Min. length: 1. Max. length: 120 | 0..1 |
| 2.9.4. Code of the organizational and legal form (csdo:BusinessEntityTypeCode) | code designation of the organizational and legal form | M.SDE.00023 | csdo:UnifiedCode20Type (M.SDT.00140) Min. length: 1. Max. length: 20 | 0..1 |
| a) identifier of the reference book (classifier) (attribute codeListId) | designation of the reference book (classifier) in accordance with which the code is specified | - | csdo:ReferenceDataIdType (M.SDT.00091) Min. length: 1. Max. length: 20 | 1 |
| 2.9.5. Name of the organizational and legal form (csdo:BusinessEntityTypeName) | name of the organizational and legal form | M.SDE.00090 | csdo:Name300Type (M.SDT.00056) Min. length: 1. Max. length: 300 | 0..1 |
| 2.9.6. Business entity identifier (csdo:BusinessEntityId) | number (code) of the record in the registry (register) assigned during state registration | M.SDE.00189 | csdo:BusinessEntityIdType (M.SDT.00157) Min. length: 1. Max. length: 20 | 0..1 |
| a) identification method (attribute kindId) | identification method of business entities | - | csdo:BusinessEntityIdKindIdType (M.SDT.00158) | 1 |
| 2.9.7. Unique customs identification number (csdo:UniqueCustomsNumberId) | unique identification number of the business entity for customs control purposes | M.SDE.00135 | csdo:UniqueCustomsNumberIdType (M.SDT.00089) Min. length: 1. Max. length: 17 | 0..1 |
| 2.9.8. Taxpayer identifier (csdo:TaxpayerId) | identifier of the business entity in the taxpayer registry | M.SDE.00025 | csdo:TaxpayerIdType (M.SDT.00025) Min. length: 1. Max. length: 20 | 0..1 |
| 2.9.9. Tax registration reason code (csdo:TaxRegistrationReasonCode) | code identifying the reason for registering the business entity for tax purposes in the Russian Federation | M.SDE.00030 | csdo:TaxRegistrationReasonCodeType (M.SDT.00030) Pattern: \d{9} | 0..1 |
| 2.9.10. Address (ccdo:SubjectAddressDetails) | address of the business entity | M.CDE.00058 | ccdo:SubjectAddressDetailsType (M.CDT.00064) Determined by the value domains of nested elements | 0..* |
| *.1. Address type code (csdo:AddressKindCode) | code designation of the address type | M.SDE.00192 | csdo:AddressKindCodeType (M.SDT.00162) Min. length: 1. Max. length: 20 | 0..1 |
| *.2. Country code (csdo:UnifiedCountryCode) | code designation of the country | M.SDE.00162 | csdo:UnifiedCountryCodeType (M.SDT.00112) Pattern: [A-Z]{2} | 0..1 |
| a) identifier of the reference book (classifier) (attribute codeListId) | designation of the reference book (classifier) | - | csdo:ReferenceDataIdType (M.SDT.00091) Min. length: 1. Max. length: 20 | 1 |
| *.3. Territory code (csdo:TerritoryCode) | code of the unit of administrative-territorial division | M.SDE.00031 | csdo:TerritoryCodeType (M.SDT.00031) Min. length: 1. Max. length: 17 | 0..1 |
| *.4. Region (csdo:RegionName) | name of the first-level administrative-territorial division unit | M.SDE.00007 | csdo:Name120Type (M.SDT.00055) Min. length: 1. Max. length: 120 | 0..1 |
| *.5. District (csdo:DistrictName) | name of the second-level administrative-territorial division unit | M.SDE.00008 | csdo:Name120Type (M.SDT.00055) Min. length: 1. Max. length: 120 | 0..1 |
| *.6. City (csdo:CityName) | name of the city | M.SDE.00009 | csdo:Name120Type (M.SDT.00055) Min. length: 1. Max. length: 120 | 0..1 |
| *.7. Settlement (csdo:SettlementName) | name of the settlement | M.SDE.00057 | csdo:Name120Type (M.SDT.00055) Min. length: 1. Max. length: 120 | 0..1 |
| *.8. Street (csdo:StreetName) | name of the element of the urban infrastructure road network | M.SDE.00010 | csdo:Name120Type (M.SDT.00055) Min. length: 1. Max. length: 120 | 0..1 |
| *.9. Building number (csdo:BuildingNumberId) | designation of the house, building, structure | M.SDE.00011 | csdo:Id50Type (M.SDT.00093) Min. length: 1. Max. length: 50 | 0..1 |
| *.10. Room number (csdo:RoomNumberId) | designation of the office or apartment | M.SDE.00012 | csdo:Id20Type (M.SDT.00092) Min. length: 1. Max. length: 20 | 0..1 |
| *.11. Post code (csdo:PostCode) | post code of the postal service enterprise | M.SDE.00006 | csdo:PostCodeType (M.SDT.00006) Pattern: [A-Z0-9][A-Z0-9-]{1,8}[A-Z0-9] | 0..1 |
| *.12. Post office box number (csdo:PostOfficeBoxId) | post office box number at the postal service enterprise | M.SDE.00013 | csdo:Id20Type (M.SDT.00092) Min. length: 1. Max. length: 20 | 0..1 |
| 2.9.11. Contact attribute (ccdo:CommunicationDetails) | contact attribute of the business entity | M.CDE.00003 | ccdo:CommunicationDetailsType (M.CDT.00003) Determined by the value domains of nested elements | 0..* |
| *.1. Communication type code (csdo:CommunicationChannelCode) | code designation of the type of communication means (channel) | M.SDE.00014 | csdo:CommunicationChannelCodeV2Type (M.SDT.00163) Min. length: 1. Max. length: 20 | 0..1 |
| *.2. Name of the communication type (csdo:CommunicationChannelName) | name of the type of communication means (channel) | M.SDE.00093 | csdo:Name120Type (M.SDT.00055) Min. length: 1. Max. length: 120 | 0..1 |
| *.3. Communication channel identifier (csdo:CommunicationChannelId) | character sequence identifying the communication channel | M.SDE.00015 | csdo:CommunicationChannelIdType (M.SDT.00015) Min. length: 1. Max. length: 1000 | 1..* |
| 2.10. Data on the authorized representative of the manufacturer of the medical device (hccdo:MedicalProductManufacturerAgentDetails) | data on the authorized representative of the manufacturer of the medical device | M.HC.CDE.00221 | ccdo:BusinessEntityDetailsType (M.CDT.00061) Determined by the value domains of nested elements | 0..1 |
| Attributes 2.10.1 - 2.10.11 have a structure similar to attributes 2.9.1 - 2.9.11 (country code, name, brief name, code of the organizational and legal form, name of the organizational and legal form, business entity identifier, unique customs identification number, taxpayer identifier, tax registration reason code, address with all sub-attributes *.1-*.12, contact attribute with all sub-attributes *.1-*.3) | ||||
| 2.11. Model name (hcsdo:ModelNumberName) | name of the model of the medical device component | M.HC.SDE.00123 | csdo:Name500Type (M.SDT.00134) Min. length: 1. Max. length: 500 | 0..1 |
| 2.12. Product identifier (csdo:ProductId) | catalog number of the medical device | M.SDE.00140 | csdo:Id50Type (M.SDT.00093) Min. length: 1. Max. length: 50 | 0..1 |
| 2.13. Code of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device (hcsdo:MedicalProductMonitoringDocKindCode) | code designation of the type of reporting document for monitoring the safety, quality, and efficacy of a medical device | M.HC.SDE.00699 | hcsdo:MedicalProductMonitoringDocKindCodeType (M.HC.SDT.00699) Possible values: 01 - report on an adverse event (incident); 02 - report on a corrective action for the safety of a medical device; 03 - report on post-registration clinical monitoring of the safety and efficacy of a medical device | 1..* |
| 2.14. Data on an adverse event (incident) (hccdo:AdverseIncidentMedicalProductDetails) | data on an adverse event (incident) | M.HC.CDE.00279 | hccdo:AdverseIncidentMedicalProductDetailsType (M.HC.CDT.00260) Determined by the value domains of nested elements | 0..1 |
| 2.14.1. Code of the manufacturer's report type on the safety of a medical device (hcsdo:ReportCorrectInfluenceTypeCode) | code designation of the manufacturer's report type on the safety of a medical device | M.HC.SDE.00547 | Possible values: 01 - initial report; 02 - subsequent report; 03 - combined initial and final report; 04 - final report | 1 |
| 2.14.2. Document number (csdo:DocId) | report number of the medical organization - user regarding the adverse event (incident) | M.SDE.00044 | csdo:Id50Type (M.SDT.00093) Min. length: 1. Max. length: 50 | 0..1 |
| 2.14.3. Document date (csdo:DocCreationDate) | report date of the medical organization - user regarding the adverse event (incident) | M.SDE.00045 | bdt:DateType (M.BDT.00005) | 1 |
| 2.14.4. Date (csdo:EventDate) | date of the adverse event (incident) | M.SDE.00131 | bdt:DateType (M.BDT.00005) | 1 |
| 2.14.5. Description (csdo:DescriptionText) | description of the adverse event (incident) | M.SDE.00002 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1 |
| 2.14.6. Date the manufacturer received information on the adverse event (incident) (hcsdo:AdverseIncidentMedicalProductManufacturingDate) | date the manufacturer received information on the adverse event (incident) | M.HC.SDE.00539 | bdt:DateType (M.BDT.00005) | 1 |
| 2.14.7. Number of patients involved (hcsdo:AdverseIncidentPatientQuantity) | number of patients involved in the adverse event (incident) | M.HC.SDE.00540 | csdo:Quantity4Type (M.SDT.00097) Max. number of digits: 4 | 0..1 |
| 2.14.8. Number of medical devices involved (hcsdo:AdverseIncidentMedicalProductQuantity) | number of medical devices involved in the adverse event (incident) | M.HC.SDE.00548 | csdo:Quantity4Type (M.SDT.00097) Max. number of digits: 4 | 0..1 |
| 2.14.9. Address (ccdo:SubjectAddressDetails) | current location of the medical device | M.CDE.00058 | ccdo:SubjectAddressDetailsType (M.CDT.00064) Determined by the value domains of nested elements (sub-attributes *.1-*.12 are similar to 2.9.10) | 0..1 |
| 2.14.10. Code of the user type of the medical device at the time of the adverse event (incident) (hcsdo:UserKindTimeAdverseIncidentCode) | code designation of the user type of the medical device at the time of the adverse event (incident) | M.HC.SDE.00542 | hcsdo:UserKindTimeAdverseIncidentCodeType (M.HC.SDT.00913) Pattern: \d{2} | 0..1 |
| 2.14.11. User of the medical device at the time of the adverse event (incident) (hcsdo:UserKindTimeAdverseIncidentName) | user of the medical device at the time of the adverse event (incident) | M.HC.SDE.00543 | csdo:Name500Type (M.SDT.00134) Min. length: 1. Max. length: 500 | 0..1 |
| 2.14.12. Code of the application type of the medical device (hcsdo:ApplicationMedicalProductCode) | code designation of the application type of the medical device | M.HC.SDE.00545 | Possible values: 01 - primary application; 02 - repeated application of a single-use medical device; 03 - repeated application of a reusable medical device; 04 - medical device after maintenance or repair; 05 - the problem was identified prior to application; 99 - other | 0..1 |
| 2.14.13. Name of the application type of the medical device (hcsdo:ApplicationMedicalProductName) | name of the application type of the medical device | M.HC.SDE.00546 | csdo:Name500Type (M.SDT.00134) Min. length: 1. Max. length: 500 | 0..1 |
| 2.15. Data on the results of the investigation of an adverse event (incident) (hccdo:ResultsInvestigationMedicalProductDetails) | data on the results of the investigation of an adverse event (incident) | M.HC.CDE.00281 | hccdo:ResultsInvestigationMedicalProductDetailsType (M.HC.CDT.00262) Determined by the value domains of nested elements | 0..1 |
| 2.15.1. Result of the analysis conducted by the manufacturer of the medical device (hcsdo:ResultInvestigationText) | description of the result of the analysis conducted by the manufacturer | M.HC.SDE.00555 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1 |
| 2.15.2. Data on the assessment of the adverse event (incident) (hccdo:IncidentAssessmentDetails) | data on the assessment of the adverse event (incident) | M.HC.CDE.00286 | Determined by the value domains of nested elements | 1..* |
| *.1. Code of the term level (hcsdo:IncidentAssessmentLevelKindCode) | code designation of the level of the term describing the adverse event (incident) | M.HC.SDE.00556 | Possible values: 01 - level 1; 02 - level 2 | 1 |
| *.2. Term code (hcsdo:IncidentAssessmentCode) | code designation of the term describing the adverse event (incident) | M.HC.SDE.00557 | hcsdo:IncidentAssessmentCodeType (M.HC.SDT.00222) Pattern: \d{4} | 0..1 |
| *.3. Term name (hcsdo:IncidentAssessmentName) | name of the term describing the adverse event (incident) | M.HC.SDE.00558 | csdo:Name500Type (M.SDT.00134) Min. length: 1. Max. length: 500 | 0..1 |
| 2.15.3. Data on the action taken based on the investigation results (hccdo:CorrectionActionPlaceDetails) | data on the action taken based on the results of the investigation of the adverse event (incident) | M.HC.CDE.00284 | Determined by the value domains of nested elements | 0..* |
| *.1. Action (hcsdo:CorrectActionPlaceText) | description of the action | M.HC.SDE.00559 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1 |
| *.2. Start date (csdo:StartDate) | start date of the action | M.SDE.00073 | bdt:DateType (M.BDT.00005) | 0..1 |
| *.3. End date (csdo:EndDate) | end date of the action | M.SDE.00074 | bdt:DateType (M.BDT.00005) | 0..1 |
| 2.15.4. Manufacturer's final comment (hcsdo:FinishCommentManufacturingText) | manufacturer's final comment based on the investigation results | M.HC.SDE.00565 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1 |
| 2.15.5. Flag of the manufacturer's awareness of similar adverse events (incidents) (hcsdo:InformedIncidentManufacturingIndicator) | flag of the manufacturer's awareness: 1 - aware; 0 - unaware | M.HC.SDE.00561 | bdt:IndicatorType (M.BDT.00013) «true» or «false» | 1 |
| 2.15.6. Data on a similar adverse event (incident) (hccdo:SimilarIncidentDetails) | data on a similar adverse event (incident) | M.HC.CDE.00287 | Determined by the value domains of nested elements | 0..* |
| *.1. Country code (csdo:UnifiedCountryCode) | code designation of the country in the territory of which the adverse event (incident) occurred | M.SDE.00162 | csdo:UnifiedCountryCodeType (M.SDT.00112) Pattern: [A-Z]{2} | 1 |
| a) identifier of the reference book (classifier) (attribute codeListId) | designation of the reference book (classifier) | - | csdo:ReferenceDataIdType (M.SDT.00091) Min. length: 1. Max. length: 20 | 1 |
| *.2. Document number (csdo:DocId) | number of the adverse event (incident) | M.SDE.00044 | csdo:Id50Type (M.SDT.00093) Min. length: 1. Max. length: 50 | 0..* |
| *.3. Number of adverse events (incidents) (hcsdo:IncidentMedicalProductQuantity) | number of adverse events (incidents) | M.HC.SDE.00562 | csdo:Quantity4Type (M.SDT.00097) Max. number of digits: 4 | 1 |
| *.4. Country code of the medical device distribution (hcsdo:DistributionUnifiedCountryCode) | code designation of the country in the territory of which the medical device was distributed | M.HC.SDE.00962 | csdo:UnifiedCountryCodeType (M.SDT.00112) Pattern: [A-Z]{2} | 0..* |
| a) identifier of the reference book (classifier) (attribute codeListId) | designation of the reference book (classifier) | - | csdo:ReferenceDataIdType (M.SDT.00091) Min. length: 1. Max. length: 20 | 1 |
| 2.16. Data on a corrective action for the safety of a medical device (hccdo:CorrectionSafetyMedicalProductDetails) | data on a corrective action for the safety of a medical device | M.HC.CDE.00280 | Determined by the value domains of nested elements | 0..* |
| 2.16.1. Report type code (hcsdo:ReportCorrectInfluenceTypeV2Code) | code of the manufacturer's report type on the corrective action | M.HC.SDE.00782 | Possible values: 01 - initial report; 02 - subsequent report; 03 - final report | 1 |
| 2.16.2. Document number (csdo:DocId) | unique identifier of the manufacturer's report on the corrective action | M.SDE.00044 | csdo:Id50Type (M.SDT.00093) Min. length: 1. Max. length: 50 | 1 |
| 2.16.3. Document date (csdo:DocCreationDate) | date of the manufacturer's report on the corrective action | M.SDE.00045 | bdt:DateType (M.BDT.00005) | 1 |
| 2.16.4. General information and reason for the corrective action (hcsdo:GeneralInformationReasonCorrectingActionText) | description of general information and reason for the corrective action | M.HC.SDE.00550 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1 |
| 2.16.5. Description and justification of the corrective action (hcsdo:DescriptionReasonCorrectingActionText) | description and justification of the corrective action | M.HC.SDE.00551 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1 |
| 2.16.6. Recommendations for suppliers (distributors) and users (hcsdo:RecommendationsText) | description of recommendations | M.HC.SDE.00511 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 0..1 |
| 2.16.7. Description of the implementation process of corrective actions (hcsdo:DescriptionRealizationProcessCorrectingActionText) | description of the implementation process of corrective actions | M.HC.SDE.00553 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 0..1 |
| 2.16.8. Data on the timing of implementation of corrective actions (hccdo:CorrectionSafetyPeriodDetails) | data on the timing of implementation of corrective actions | M.HC.CDE.00895 | Determined by the value domains of nested elements | 0..1 |
| *.1. Start date (csdo:StartDate) | start date of the implementation of the corrective action | M.SDE.00073 | bdt:DateType (M.BDT.00005) | 1 |
| *.2. End date (csdo:EndDate) | end date of the implementation of the corrective action | M.SDE.00074 | bdt:DateType (M.BDT.00005) | 1 |
| 2.17. Data on post-registration clinical monitoring of the safety and efficacy of a medical device (hcsdo:PostRegisrationClinicalMonitoringMedicalProductDetails) | data on post-registration clinical monitoring | M.HC.CDE.00282 | Determined by the value domains of nested elements | 0..1 |
| 2.17.1. Report type code (hcsdo:ReportCorrectInfluenceTypeV2Code) | code of the manufacturer's report type on post-registration clinical monitoring | M.HC.SDE.00782 | Possible values: 01 - initial report; 02 - subsequent report; 03 - final report | 1 |
| 2.17.2. Document number (csdo:DocId) | unique identifier of the manufacturer's report on post-registration clinical monitoring | M.SDE.00044 | csdo:Id50Type (M.SDT.00093) Min. length: 1. Max. length: 50 | 1 |
| 2.17.3. Document date (csdo:DocCreationDate) | date of the manufacturer's report on post-registration clinical monitoring | M.SDE.00045 | bdt:DateType (M.BDT.00005) | 1 |
| 2.17.4. Identified residual risk associated with the medical device (hcsdo:ListIdentificationResidualRiskText) | description of the identified residual risk | M.HC.SDE.00566 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1..* |
| 2.17.5. Goal and objective of the post-registration clinical monitoring (hcsdo:PurposesPostRegistrationClinicalMonitoringText) | description of the goal and objective of the post-registration clinical monitoring | M.HC.SDE.00568 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1..* |
| 2.17.6. Data on the scheme of post-registration clinical monitoring (hccdo:SchemePostRegistrationClinicalMonitoringDetails) | data on the scheme of post-registration clinical monitoring | M.HC.CDE.00819 | Determined by the value domains of nested elements | 1 |
| *.1. Scheme of post-registration clinical monitoring (hcsdo:SchemePostRegistrationClinicalMonitoringText) | description of the scheme | M.HC.SDE.00580 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 0..1 |
| *.2. Document in PDF format (hcsdo:PdfBinaryText) | document in PDF format | M.HC.SDE.00326 | csdo:BinaryTextType (M.SDT.00143) Finite sequence of binary octets (bytes) | 0..1 |
| a) data format code (attribute mediaTypeCode) | code designation of the data format | - | csdo:MediaTypeCodeType (M.SDT.00147) Min. length: 1. Max. length: 255 | 0..1 |
| 2.17.7. Data on clinical data obtained during the reporting period (hccdo:ClinicalDataDetails) | data on clinical data obtained during the reporting period | M.HC.CDE.00820 | Determined by the value domains of nested elements | 1 |
| *.1. Clinical data obtained during the reporting period (hcsdo:ClinicalText) | description of clinical data | M.HC.SDE.00570 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 0..1 |
| *.2. Document in PDF format (hcsdo:PdfBinaryText) | document in PDF format | M.HC.SDE.00326 | csdo:BinaryTextType (M.SDT.00143) | 0..1 |
| a) data format code (attribute mediaTypeCode) | code designation of the data format | - | csdo:MediaTypeCodeType (M.SDT.00147) Min. length: 1. Max. length: 255 | 0..1 |
| 2.17.8. Assessment of clinical data obtained during the reporting period (hcsdo:MarketClinicalText) | description of the assessment of clinical data for the reporting period | M.HC.SDE.00571 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 0..1 |
| 2.17.9. Assessment of clinical data obtained during the period of post-registration clinical monitoring (hcsdo:MarketClinicalAllText) | description of the assessment of clinical data for the period of post-registration clinical monitoring | M.HC.SDE.00572 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1 |
| 2.17.10. Conclusion on the need (absence of need) to adjust the post-registration clinical monitoring plan (hcsdo:ConclusionNeedPostRegistrationClinicalMonitoringText) | description of the conclusion on the need to adjust the plan | M.HC.SDE.00574 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1 |
| 2.17.11. Conclusion on the need (absence of need) to perform corrective actions regarding the safety of the medical device (hcsdo:ConclusionNeedPostRegistrationClinicalMonitoringActionText) | description of the conclusion on the need for corrective actions | M.HC.SDE.00579 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1 |
| 2.17.12. Description of corrective actions (hcsdo:DescriptionPostRegistrationClinicalMonitoringActionText) | description of corrective actions | M.HC.SDE.00576 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 0..1 |
| 2.17.13. Conclusion (justification) on the clinical safety and efficacy of the medical device (hcsdo:ConclusionNeedSafetyText) | description of the conclusion on clinical safety and efficacy | M.HC.SDE.00578 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1 |
| 2.17.14. Conclusion on the need (absence of need) to extend the post-registration clinical monitoring cycle (hcsdo:ConclusionNeedPostRegistrationClinicalMonitoringCycleText) | description of the conclusion on the need to extend the cycle | M.HC.SDE.00575 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 1 |
| 2.17.15. Comment on post-registration clinical monitoring (hcsdo:PostRegisrationClinicalMonitoringMedicalProductCommentText) | description of the comment on post-registration clinical monitoring | M.HC.SDE.00589 | csdo:Text4000Type (M.SDT.00088) Min. length: 1. Max. length: 4000 | 0..1 |
| 2.18. Document in PDF format (hcsdo:PdfBinaryText) | copy of the manufacturer's notification on the safety of the medical device | M.HC.SDE.00326 | csdo:BinaryTextType (M.SDT.00143) Finite sequence of binary octets (bytes) | 0..1 |
| a) data format code (attribute mediaTypeCode) | code designation of the data format | - | csdo:MediaTypeCodeType (M.SDT.00147) Min. length: 1. Max. length: 255 | 0..1 |
| 2.19. Technological characteristics of the shared resource record (ccdo:ResourceItemStatusDetails) | set of technological data about the shared resource record | M.CDE.00032 | ccdo:ResourceItemStatusDetailsType (M.CDT.00033) Determined by the value domains of nested elements | 1 |
| 2.19.1. Validity period (ccdo:ValidityPeriodDetails) | validity period of the shared resource record (register, list, database) | M.CDE.00033 | ccdo:PeriodDetailsType (M.CDT.00026) Determined by the value domains of nested elements | 0..1 |
| *.1. Start date and time (csdo:StartDateTime) | start date and time | M.SDE.00133 | bdt:DateTimeType (M.BDT.00006) Designation of date and time in accordance with GOST ISO 8601-2001 | 0..1 |
| *.2. End date and time (csdo:EndDateTime) | end date and time | M.SDE.00134 | bdt:DateTimeType (M.BDT.00006) Designation of date and time in accordance with GOST ISO 8601-2001 | 0..1 |
| 2.19.2. Date and time of update (csdo:UpdateDateTime) | date and time of updating of the shared resource record (register, list, database) | M.SDE.00079 | bdt:DateTimeType (M.BDT.00006) Designation of date and time in accordance with GOST ISO 8601-2001 | 0..1 |